Phenylpropanoids metabolism
The biosynthesis of phenylpropanoids involves a number of enzymes.
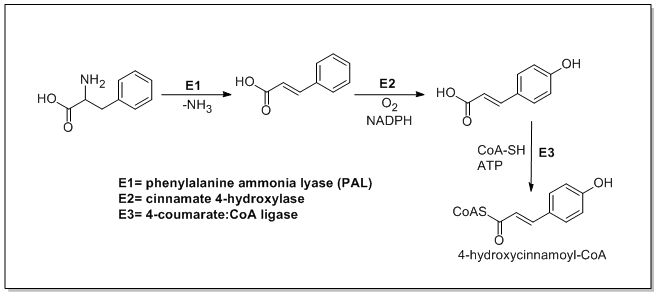
From amino acids to cinnamates
In plants, all phenylpropanoids are derived from the amino acids phenylalanine and tyrosine.
p-coumaric acid
, respectively.
4-coumaroyl-CoA.[1]
Enzymes associated with biosynthesis of hydroxycinnamic acids
- Cinnamyl-alcohol dehydrogenase (CAD), an enzyme that transforms cinnamyl alcohol into cinnamaldehyde
- sinapic acid) and choline
- 2-hydroxycinnamate
- Caffeate O-methyltransferase, an enzyme that transforms caffeic acid into ferulic acid
- Caffeoyl-CoA O-methyltransferase, an enzyme that transforms caffeoyl-CoA into feruloyl-CoA
- 5-O-(4-coumaroyl)-D-quinate 3'-monooxygenase, an enzyme that transforms trans-5-O-(4-coumaroyl)-D-quinate into trans-5-O-caffeoyl-D-quinate
- Sinapoylglucose—choline O-sinapoyltransferase, an enzyme that transforms 1-O-sinapoyl-beta-D-glucose into sinapoylcholine (sinapine)
- Sinapoylglucose—malate O-sinapoyltransferase, an enzyme that transforms 1-O-sinapoyl-beta-D-glucose into sinapoyl-(S)-malate
- Cinnamoyl-CoA reductase, an enzyme that transforms cinnamoyl-CoA from cinnamaldehyde
Conjugation enzymes
These enzymes
conjugate
phenylpropanoids to other molecules.
- trans-2-hydroxycinnamate into trans-beta-D-glucosyl-2-hydroxycinnamate
- 4-O-beta-D-glucosyl-4-hydroxycinnamate
- 4-coumaroyl-CoA into 4-coumaroylshikimate
- Quinate O-hydroxycinnamoyltransferase, the enzyme that transforms feruloyl-CoA into O-feruloylquinate
- sinapic acid) into 1-sinapoyl-D-glucose
- Coniferyl-alcohol glucosyltransferase, the enzyme that transforms coniferyl alcohol into coniferin
Deconjugation enzymes
- coniferol
Stilbenoids biosynthesis
- Pinosylvin synthase, an enzyme that transforms pinosylvin from cinnamoyl-CoA
- 4-coumaroyl-CoA to resveratrol.
An alternative bacterial
3,5-dihydroxy-4-isopropyl-trans-stilbene for antibiotic purposes.[2]
Coumarins biosynthesis
- Scopoletin glucosyltransferase, the enzyme that transforms scopoletin into scopolin
Chalcones biosynthesis
Naringenin-chalcone synthase
is an enzyme that catalyzes the following conversion:
- 3-malonyl-CoA + 4-coumaroyl-CoA → 4 CoA + naringenin chalcone + 3 CO2
Flavonoids biosynthesis
Conjugate ring-closure of chalcones results in the familiar form of flavonoids, the three-ringed structure of a flavone.
Biodegradation
Hydroxycinnamic acids degradation
- Caffeate 3,4-dioxygenase is an enzyme that uses 3,4-dihydroxy-trans-cinnamate (caffeic acid) and oxygen to produce 3-(2-carboxyethenyl)-cis,cis-muconate.