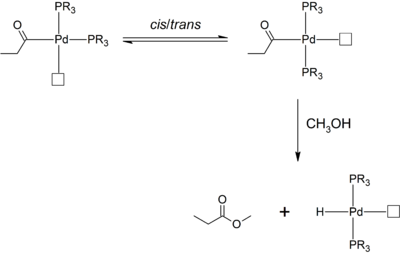Polyketone

Polyketones are a family of high-performance
Industrial production
The ethylene-carbon monoxide co-polymer is most significant. Industrially, this polymer is synthesized either as a methanol slurry, or via a gas phase reaction with immobilized catalysts.[4][5]
Polymerization mechanism
Initiation and termination
Where external initiation is not employed for the methanol system, initiation can take place via
Propagation
A mechanism for the propagation of this reaction using a palladium(II)-
Polyketones are noted for having extremely low defects (double ethylene insertions or double carbonyl insertions, in red):
The activation barrier to give double carbonyl insertions is very high, so it does not occur.[6] Brookhart's mechanistic studies show that the concentration of the alkyl-ethylene palladium complex required to give double ethylene insertions is very low at any one point:
Additionally, the Gibbs energy of activation of the alkyl-ethylene insertion is ~ 3 kcal/mol higher than the corresponding activation barrier for the alkyl-carbon monoxide insertion. As a result, defects occur at an extremely low rate (~ 1 part per million).
Importance of bidentate ligands
Where palladium(II) pre-catalysts bearing monodentate phosphine ligands are used in methanol, a relatively high fraction of methyl propionate is produced. In comparison, where chelating
Whereas much effort has involved discrete palladium complexes, an example in the patent literature claims that a combination of
References
- ^ "FindArticles.com | CBSi". findarticles.com. Retrieved 2021-05-24.
- ^ "Shell Carilon® DP P1000 Polyketone". www.matweb.com. Retrieved 2021-05-24.
- ^ Vink, David (15 September 2014). "Finding openings for polyketone compounds". PlasticsNewsEurope. Archived from the original on 18 May 2015. Retrieved 12 May 2015.
- ISBN 9781118633892.
- .
- ^ PMID 11848769.
- ^ .
- .
- ^ Grant Proulx, (2000), "Preparation of olefin copolymers of sulfur dioxide or carbon monoxide", US Patent US006037442A