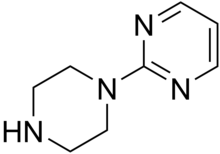
Pyrimidinylpiperazine
| |
| Names | |
|---|---|
| Preferred IUPAC name
2-(Piperazin-1-yl)pyrimidine | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.040.107 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H12N4 | |
| Molar mass | 164.21 g/mol |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H314, H315, H319, H335 | |
| P260, P261, P264, P271, P280, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P332+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
1-(2-Pyrimidinyl)piperazine (1-PP, 1-PmP) is a
α1-adrenergic receptors.[4][additional citation(s) needed] Its crystal structure has been determined.[5]
Derivatives
A number of pyrimidinylpiperazine derivatives are drugs, including:
- Buspirone – anxiolytic
- Dasatinib – anticancer agent
- Eptapirone – anxiolytic
- Gepirone – anxiolytic
- Ipsapirone – anxiolytic
- Piribedil – antiparkinsonian agent
- Revospirone – anxiolytic
- Tandospirone – anxiolytic
- Tirilazad – neuroprotective agent
- Umespirone – anxiolytic
- Zalospirone – anxiolytic
The anxiolytics are also classified as azapirones due to the azaspirodecanedione moiety in their structures. 1-PP is a common metabolite of most or all of the listed agents.[1][6] Alnespirone, binospirone, and enilospirone, despite being azapirones, are not piperazines and therefore do not metabolize to 1-PP, and while perospirone and tiospirone are piperazines, they are instead benzothiazole-substituted piperazines and do not metabolize to 1-PP either.
See also
- Substituted piperazine
- Pyridinylpiperazine
- Phenylpiperazine
- Diphenylmethylpiperazine
- Benzylpiperazine
