Reaction–diffusion system

Reaction–diffusion systems are mathematical models that correspond to several physical phenomena. The most common is the change in space and time of the concentration of one or more chemical substances: local chemical reactions in which the substances are transformed into each other, and diffusion which causes the substances to spread out over a surface in space.
Reaction–diffusion systems are naturally applied in chemistry. However, the system can also describe dynamical processes of non-chemical nature. Examples are found in biology, geology and physics (neutron diffusion theory) and ecology. Mathematically, reaction–diffusion systems take the form of semi-linear parabolic partial differential equations. They can be represented in the general form
where q(x, t) represents the unknown vector function, D is a
One-component reaction–diffusion equations
The simplest reaction–diffusion equation is in one spatial dimension in plane geometry,
is also referred to as the
The dynamics of one-component systems is subject to certain restrictions as the evolution equation can also be written in the variational form
and therefore describes a permanent decrease of the "free energy" given by the functional
with a potential V(u) such that R(u) = dV(u)/du.

In systems with more than one stationary homogeneous solution, a typical solution is given by travelling fronts connecting the homogeneous states. These solutions move with constant speed without changing their shape and are of the form u(x, t) = û(ξ) with ξ = x − ct, where c is the speed of the travelling wave. Note that while travelling waves are generically stable structures, all non-monotonous stationary solutions (e.g. localized domains composed of a front-antifront pair) are unstable. For c = 0, there is a simple proof for this statement:[8] if u0(x) is a stationary solution and u = u0(x) + ũ(x, t) is an infinitesimally perturbed solution, linear stability analysis yields the equation
With the ansatz ũ = ψ(x)exp(−λt) we arrive at the eigenvalue problem
of
To determine the velocity c of a moving front, one may go to a moving coordinate system and look at stationary solutions:
This equation has a nice mechanical analogue as the motion of a mass D with position û in the course of the "time" ξ under the force R with the damping coefficient c which allows for a rather illustrative access to the construction of different types of solutions and the determination of c.
When going from one to more space dimensions, a number of statements from one-dimensional systems can still be applied. Planar or curved wave fronts are typical structures, and a new effect arises as the local velocity of a curved front becomes dependent on the local
Two-component reaction–diffusion equations
Two-component systems allow for a much larger range of possible phenomena than their one-component counterparts. An important idea that was first proposed by Alan Turing is that a state that is stable in the local system can become unstable in the presence of diffusion.[10]
A linear stability analysis however shows that when linearizing the general two-component system
a plane wave perturbation
of the stationary homogeneous solution will satisfy
Turing's idea can only be realized in four equivalence classes of systems characterized by the signs of the Jacobian R′ of the reaction function. In particular, if a finite wave vector k is supposed to be the most unstable one, the Jacobian must have the signs
This class of systems is named activator-inhibitor system after its first representative: close to the ground state, one component stimulates the production of both components while the other one inhibits their growth. Its most prominent representative is the
with f (u) = λu − u3 − κ which describes how an action potential travels through a nerve.[11][12] Here, du, dv, τ, σ and λ are positive constants.
When an activator-inhibitor system undergoes a change of parameters, one may pass from conditions under which a homogeneous ground state is stable to conditions under which it is linearly unstable. The corresponding bifurcation may be either a Hopf bifurcation to a globally oscillating homogeneous state with a dominant wave number k = 0 or a Turing bifurcation to a globally patterned state with a dominant finite wave number. The latter in two spatial dimensions typically leads to stripe or hexagonal patterns.
- Subcritical Turing bifurcation: formation of a hexagonal pattern from noisy initial conditions in the above two-component reaction–diffusion system of Fitzhugh–Nagumo type.
-
Noisy initial conditions at t = 0.
-
State of the system at t = 10.
-
Almost converged state at t = 100.
For the Fitzhugh–Nagumo example, the neutral stability curves marking the boundary of the linearly stable region for the Turing and Hopf bifurcation are given by
If the bifurcation is subcritical, often localized structures (
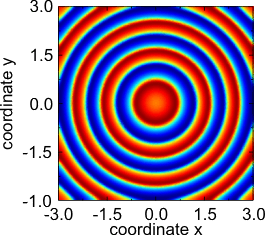
- Other patterns found in the above two-component reaction–diffusion system of Fitzhugh–Nagumo type.
-
Rotating spiral.
-
Target pattern.
-
Stationary localized pulse (dissipative soliton).
Three- and more-component reaction–diffusion equations
For a variety of systems, reaction–diffusion equations with more than two components have been proposed, e.g. the
It is known that systems with more components allow for a variety of phenomena not possible in systems with one or two components (e.g. stable running pulses in more than one spatial dimension without global feedback).[18] An introduction and systematic overview of the possible phenomena in dependence on the properties of the underlying system is given in.[19]
Applications and universality
In recent times, reaction–diffusion systems have attracted much interest as a prototype model for pattern formation.[20] The above-mentioned patterns (fronts, spirals, targets, hexagons, stripes and dissipative solitons) can be found in various types of reaction–diffusion systems in spite of large discrepancies e.g. in the local reaction terms. It has also been argued that reaction–diffusion processes are an essential basis for processes connected to morphogenesis in biology[21][22] and may even be related to animal coats and skin pigmentation.[23][24] Other applications of reaction–diffusion equations include ecological invasions,[25] spread of epidemics,[26] tumour growth,[27][28][29] dynamics of fission waves,[30] wound healing[31] and visual hallucinations.[32] Another reason for the interest in reaction–diffusion systems is that although they are nonlinear partial differential equations, there are often possibilities for an analytical treatment.[8][9][33][34][35][20]
Experiments
Well-controllable experiments in chemical reaction–diffusion systems have up to now been realized in three ways. First, gel reactors[36] or filled capillary tubes[37] may be used. Second, temperature pulses on catalytic surfaces have been investigated.[38][39] Third, the propagation of running nerve pulses is modelled using reaction–diffusion systems.[11][40]
Aside from these generic examples, it has turned out that under appropriate circumstances electric transport systems like plasmas[41] or semiconductors[42] can be described in a reaction–diffusion approach. For these systems various experiments on pattern formation have been carried out.
Numerical treatments
A reaction–diffusion system can be solved by using methods of
See also
- Autowave
- Diffusion-controlled reaction
- Chemical kinetics
- Phase space method
- Autocatalytic reactions and order creation
- Pattern formation
- Patterns in nature
- Periodic travelling wave
- Stochastic geometry
- MClone
- The Chemical Basis of Morphogenesis
- Turing pattern
- Multi-state modeling of biomolecules
Examples
- Fisher's equation
- Zeldovich–Frank-Kamenetskii equation
- FitzHugh–Nagumo model
- Wrinkle paint
References
- ISBN 978-0198747826.
- ^ Kolmogorov, A., Petrovskii, I. and Piskunov, N. (1937) Study of a Diffusion Equation That Is Related to the Growth of a Quality of Matter and Its Application to a Biological Problem. Moscow University Mathematics Bulletin, 1, 1-26.
- ^ R. A. Fisher, Ann. Eug. 7 (1937): 355
- S2CID 73620481.
- S2CID 122764449.
- ^ Y. B. Zeldovich and D. A. Frank-Kamenetsky, Acta Physicochim. 9 (1938): 341
- ^ B. H. Gilding and R. Kersner, Travelling Waves in Nonlinear Diffusion Convection Reaction, Birkhäuser (2004)
- ^ a b P. C. Fife, Mathematical Aspects of Reacting and Diffusing Systems, Springer (1979)
- ^ a b A. S. Mikhailov, Foundations of Synergetics I. Distributed Active Systems, Springer (1990)
- ISSN 2054-0280.
- ^ PMID 19431309.
- ^ J. Nagumo et al., Proc. Inst. Radio Engin. Electr. 50 (1962): 2061
- ISSN 0022-2526.
- PMID 15089714.
- PMID 15447151.
- PMID 22757555.
- ^ H.-G. Purwins et al. in: Dissipative Solitons, Lectures Notes in Physics, Ed. N. Akhmediev and A. Ankiewicz, Springer (2005)
- ISSN 0031-9007.
- ISBN 978-3-642-31250-2
- ^ ISSN 1385-8947.
- ^ L.G. Harrison, Kinetic Theory of Living Pattern, Cambridge University Press (1993)
- PMID 33449631.
- ^ H. Meinhardt, Models of Biological Pattern Formation, Academic Press (1982)
- ISBN 978-3-662-08539-4.
- S2CID 85421773.
- S2CID 129301761.
- ISSN 0218-3390.
- S2CID 11967813.
- ^ R.A. Gatenby and E.T. Gawlinski, Cancer Res. 56 (1996): 5745
- S2CID 240589650.
- S2CID 20717487.
- ^ https://www.quantamagazine.org/a-math-theory-for-why-people-hallucinate-20180730/
- ^ P. Grindrod, Patterns and Waves: The Theory and Applications of Reaction-Diffusion Equations, Clarendon Press (1991)
- ^ J. Smoller, Shock Waves and Reaction Diffusion Equations, Springer (1994)
- ^ B. S. Kerner and V. V. Osipov, Autosolitons. A New Approach to Problems of Self-Organization and Turbulence, Kluwer Academic Publishers (1994)
- S2CID 4257570.
- ISSN 1367-2630.
- PMID 10043694.
- ISSN 0022-3654.
- PMID 12991237.
- ISSN 0167-2789.
- ^ E. Schöll, Nonlinear Spatio-Temporal Dynamics and Chaos in Semiconductors, Cambridge University Press (2001)
- ^ S.Tang et al., J.Austral.Math.Soc. Ser.B 35(1993): 223–243
- ^ Tim Hutton, Robert Munafo, Andrew Trevorrow, Tom Rokicki, Dan Wills. "Ready, a cross-platform implementation of various reaction-diffusion systems." https://github.com/GollyGang/ready
- .
- ^ Linker, Patrick (2016). "Numerical methods for solving the reactive diffusion equation in complex geometries". The Winnower.
- ^ Simulation tools for particle-based reaction-diffusion dynamics in continuous space https://link.springer.com/article/10.1186/s13628-014-0011-5
- ^ Fröhner, Christoph, and Frank Noé. "Reversible interacting-particle reaction dynamics." The Journal of Physical Chemistry B 122.49 (2018): 11240-11250.
External links
- Reaction–Diffusion by the Gray–Scott Model: Pearson's parameterization a visual map of the parameter space of Gray–Scott reaction diffusion.
- A thesis on reaction–diffusion patterns with an overview of the field
- RD Tool: an interactive web application for reaction-diffusion simulation





![{\displaystyle {\mathfrak {L}}=\int _{-\infty }^{\infty }\left[{\tfrac {D}{2}}\left(\partial _{x}u\right)^{2}-V(u)\right]\,{\text{d}}x}](https://wikimedia.org/api/rest_v1/media/math/render/svg/ae3c05e847595f155e3a7cb073798c6d01220d3a)











![{\displaystyle {\begin{aligned}q_{\text{n}}^{H}(k):&{}\quad {\frac {1}{\tau }}+\left(d_{u}^{2}+{\frac {1}{\tau }}d_{v}^{2}\right)k^{2}&=f^{\prime }(u_{h}),\\[6pt]q_{\text{n}}^{T}(k):&{}\quad {\frac {\kappa }{1+d_{v}^{2}k^{2}}}+d_{u}^{2}k^{2}&=f^{\prime }(u_{h}).\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/44e1fc5b936101f66caea7f346e2c29a3b879ef2)