Sarcopenia
| Sarcopenia | |
|---|---|
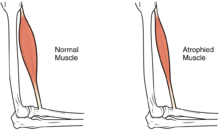 | |
| Difference between a normal muscle and an atrophied muscle | |
| Specialty | Geriatrics Rheumatology |
Sarcopenia (
Sarcopenia is a factor in changing body composition. When associated with aging populations, certain muscle regions are expected to be affected first, specifically the anterior thigh and abdominal muscles.[2][4] In population studies, body mass index (BMI) is seen to decrease in aging populations while bioelectrical impedance analysis (BIA) shows body fat proportion rising.[5]
Etymology
The term sarcopenia stems from Greek σάρξ sarx, "flesh" and πενία penia, "poverty". This was first proposed by Rosenberg in 1989, who wrote that "there may be no single feature of age-related decline that could more dramatically affect ambulation, mobility, calorie intake, and overall nutrient intake and status, independence, breathing, etc".[citation needed]
Sarcopenia is distinct from cachexia, in which muscle is degraded through cytokine-mediated degradation, although the two conditions may co-exist.
Signs and symptoms
The hallmark sign of sarcopenia is loss of lean muscle mass, or muscle atrophy. The change in body composition may be difficult to detect due to obesity, changes in fat mass, or edema. Changes in weight, limb or waist circumference are not reliable indicators of muscle mass changes. Sarcopenia may also cause reduced strength, functional decline and increased risk of falling. Sarcopenia may also have no symptoms until it is severe and is often unrecognized.[1] Research has shown, however, that hypertrophy may occur in the upper parts of the body to compensate for this loss of lean muscle mass[2][6] Therefore, one early indicator of the onset of sarcopenia can be significant loss of muscle mass in the anterior thigh and abdominal muscles.[2]
Causes
There are many proposed causes of sarcopenia and it is likely the result of multiple interacting factors. Understanding of the causes of sarcopenia is incomplete, however changes in hormones, immobility, age-related muscle changes, nutrition and neurodegenerative changes have all been recognized as potential causative factors.[7]
The degree of sarcopenia is determined by two factors: the initial amount of muscle mass and the rate at which muscle mass declines. Due to variations in these factors across the population, the rate of progression and the threshold at which muscle loss becomes apparent is variable.[8] Immobility dramatically increases the rate of muscle loss, even in younger people. Other factors that can increase the rate of progression of sarcopenia include decreased nutrient intake, low physical activity, or chronic disease.[1] Additionally, epidemiological research has indicated that early environmental influences may have long-term effects on muscle health. For example, low birth weight, a marker of a poor early environment, is associated with reduced muscle mass and strength in adult life.[9][10][11]
Pathophysiology
There are multiple theories proposed to explain the mechanisms of muscle changes of sarcopenia including changes in myosatellite cell recruitment, changes in anabolic signalling, protein oxidation, inflammation, and developmental factors. The pathologic changes of sarcopenia include a reduction in muscle tissue quality as reflected in the replacement of muscle fibers with fat, an increase in fibrosis, changes in muscle metabolism, oxidative stress, and degeneration of the neuromuscular junction.[12] The failure to activate satellite cells upon injury or exercise is also thought to contribute to the pathophysiology of sarcopenia.[12] Additionally, oxidized proteins can lead to a buildup of lipofuscin and cross-linked proteins causing an accumulation of non-contractile material in the skeletal muscle and contribute to sarcopenic muscle.[8]
In sarcopenic muscle the distribution of the types of
Aging sarcopenic muscle shows an accumulation of
Furthermore, not every muscle is as susceptible to the atrophic effects of aging. For example, in both humans[18] and mice[19] it has been shown that lower leg muscles are not as susceptible to aging as upper leg muscles. This could perhaps be explained by the differential distribution of myofiber type within each muscle group, but this is unknown.
Diagnosis
Multiple diagnostic criteria have been proposed by various expert groups and continues to be an area of research and debate. Despite the lack of a widely accepted definition, sarcopenia was assigned an ICD-10 code (M62.84) in 2016, recognizing it as a disease state.[20]
Sarcopenia can be diagnosed when a patient has muscle mass that is at least two
There are screening tools for sarcopenia that assess patient reported difficulty in doing daily activities such as walking, climbing stairs or standing from a chair and have been shown to predict sarcopenia and poor functional outcomes.[23]
Management
Exercise
Exercise remains the intervention of choice for sarcopenia, but translation of research findings into clinical practice is challenging. The type, duration and intensity of exercise are variable between studies, preventing a standardized exercise prescription for sarcopenia.[24] Lack of exercise is a significant risk factor for sarcopenia and exercise can dramatically slow the rate of muscle loss.[25] Exercise can be an effective intervention because aging skeletal muscle retains the ability to synthesize proteins in response to short-term resistance exercise.[26] Progressive resistance training in older adults can improve physical performance (gait speed) and muscular strength.[27] Increased exercise can produce greater numbers of cellular mitochondria, increase capillary density, and increase the mass and strength of connective tissue.[28]
Medication
There are currently no approved medications for the treatment of sarcopenia.[29] Testosterone or other anabolic steroids have also been investigated for treatment of sarcopenia, and seem to have some positive effects on muscle strength and mass, but cause several side effects and raise concerns of prostate cancer in men and virilization in women.[30][31] Additionally, recent studies suggest testosterone treatments may induce adverse cardiovascular events.[32][33][34]
Other medications under investigation as possible treatments for sarcopenia include
Nutrition
Intake of calories and protein are important stimuli for muscle protein synthesis.[35] Older adults may not utilize protein as efficiently as younger people and may require higher amounts to prevent muscle atrophy.[21] A number of expert groups have proposed an increase in dietary protein recommendations for older age groups to 1.0–1.2 g/kg body weight per day.[36][37] Ensuring adequate nutrition in older adults is of interest in the prevention of sarcopenia and frailty, since it is a simple, low-cost treatment approach without major side effects.[38]
Supplements
A component of sarcopenia is the loss of ability for aging skeletal muscle to respond to anabolic stimuli such as
Epidemiology
The prevalence of sarcopenia depends on the definition used in each epidemiological study. Estimated prevalence in people between the ages of 60-70 is 5-13% and increases to 11-50% in people more than 80 years of age. This equates to >50 million people and is projected to affect >200 million in the next 40 years given the rising population of older adults.[7]
Public health impact
Sarcopenia is emerging as a major public health concern given the increased longevity of industrialized populations and growing geriatric population. Sarcopenia is a predictor of many adverse outcomes including increased disability, falls and mortality.[43][44] Immobility or bed rest in populations predisposed to sarcopenia can cause dramatic impact on functional outcomes. In the elderly, this often leads to decreased biological reserve and increased vulnerability to stressors known as the "frailty syndrome". Loss of lean body mass is also associated with increased risk of infection, decreased immunity, and poor wound healing. The weakness that accompanies muscle atrophy leads to higher risk of falls, fractures, physical disability, need for institutional care, reduced quality of life, increased mortality, and increased healthcare costs.[21] This represents a significant personal and societal burden and its public health impact is increasingly recognized.[7]
Research directions
There are significant opportunities to better understand the causes and consequences of sarcopenia and help guide clinical care. This includes elucidation of the molecular and cellular mechanisms of sarcopenia, further refinement of reference populations by ethnic groups, validation of diagnostic criteria and clinical tools, as well as tracking of incidence of hospitalization admissions, morbidity, and mortality. Identification and research on potential therapeutic approaches and timing of interventions is also needed.[45]
As of 2020[update], there are no drugs approved to treat muscle wasting in people with chronic diseases, and there is therefore an unmet need for anabolic drugs with few side effects. One aspect hindering drug approval for treatments for cachexia and sarcopenia is disagreement in endpoints. Several clinical trials have found that
See also
- Ageing – Biological process of getting older
- Cachexia – Syndrome causing muscle loss not entirely reversible
- Dynapenia – Loss of muscular strength not caused by neurological or muscular disease
- Frailty syndrome – Weakness in elderly person
- Geriatrics – Specialty that focuses on health care of elderly people
References
- ^ S2CID 206555460.
- ^ S2CID 211245329.
- PMID 28095426.
- S2CID 96463073.
- PMID 24004464.
- PMID 31268513.
- ^ PMID 20392703.
- ^ PMID 14570858.
- PMID 15472158.
- PMID 11232011.
- PMID 17356523.
- ^ S2CID 8576449.
- PMID 12970377.
- PMID 36820956.
- S2CID 21609241.
- PMID 36442091.
- PMID 35263007.
- PMID 36825643.
- PMC 11009184.
- ^ PMID 27891296.
- ^ PMID 27324808.
- S2CID 220113690.
- PMID 27066316.
- PMID 25227204.
- PMID 17117147.
- PMID 14570859.
- PMID 19588334.
- ISBN 978-1-947172-04-3.
- ^ PMID 26178029.
- ^ PMID 22690213.
- ^ PMID 24219006.
- PMID 24489673.
- PMID 24193080.
- PMID 20592293.
- PMID 28927897.
- PMID 23867520.
- PMID 24814383.
- PMID 30115829.
- PMID 16365098.
- S2CID 29717535.
- ^ PMID 26169182.
- PMID 28493406.
- PMID 32924316.
- PMID 35055695.
- PMID 23315797.
- S2CID 259232987.
- S2CID 219174372.
- PMID 32257854.
Further reading
- Fujita S, Volpi E (January 2006). "Amino acids and muscle loss with aging". The Journal of Nutrition (Review). 136 (1 Suppl): 277S–280S. PMID 16365098.
- Roubenoff R (December 2007). "Physical activity, inflammation, and muscle loss". Nutrition Reviews. 65 (12 Pt 2): S208–S212. PMID 18240550.
- Sharlo K, Tyganov SA, Tomilovskaya E, Popov DV, Saveko AA, Shenkman BS (December 2021). "Effects of Various Muscle Disuse States and Countermeasures on Muscle Molecular Signaling". International Journal of Molecular Sciences. 23 (1): 468. PMID 35008893.
