Spindle apparatus

In
Besides chromosomes, the spindle apparatus is composed of hundreds of
Spindle structure
Attachment of microtubules to chromosomes is mediated by
The cellular spindle apparatus includes the spindle microtubules, associated proteins, which include kinesin and dynein molecular motors, condensed chromosomes, and any centrosomes or asters that may be present at the spindle poles depending on the cell type.[4] The spindle apparatus is vaguely ellipsoid in cross section and tapers at the ends. In the wide middle portion, known as the spindle midzone, antiparallel microtubules are bundled by kinesins. At the pointed ends, known as spindle poles, microtubules are nucleated by the centrosomes in most animal cells. Acentrosomal or anastral spindles lack centrosomes or asters at the spindle poles, respectively, and occur for example during female meiosis in most animals.[5] In this instance, a Ran GTP gradient is the main regulator of spindle microtubule organization and assembly. In fungi, spindles form between spindle pole bodies embedded in the nuclear envelope, which does not break down during mitosis.
Microtubule-associated proteins and spindle dynamics
The dynamic lengthening and shortening of spindle microtubules, through a process known as
The growing ends of microtubules are protected against catastrophe by the action of plus-end microtubule tracking proteins (+TIPs) to promote their association with kinetochores at the midzone.
Opposing the action of these microtubule-stabilizing proteins are a number of microtubule-depolymerizing factors which permit the dynamic remodeling of the mitotic spindle to promote chromosome congression and attainment of bipolarity. The kinesin-13 superfamily of MAPs contains a class of plus-end-directed motor proteins with associated microtubule depolymerization activity including the well-studied mammalian MCAK and Xenopus XKCM1. MCAK localizes to the growing tips of microtubules at kinetochores where it can trigger catastrophe in direct competition with stabilizing +TIP activity.[15] These proteins harness the energy of ATP hydrolysis to induce destabilizing conformational changes in protofilament structure that cause kinesin release and microtubule depolymerization.[16] Loss of their activity results in numerous mitotic defects.[15] Additional microtubule destabilizing proteins include Op18/stathmin and katanin which have roles in remodeling the mitotic spindle as well as promoting chromosome segregation during anaphase.[17]
The activities of these MAPs are carefully regulated to maintain proper microtubule dynamics during spindle assembly, with many of these proteins serving as Aurora and Polo-like kinase substrates.[17][18]
Organizing the spindle apparatus
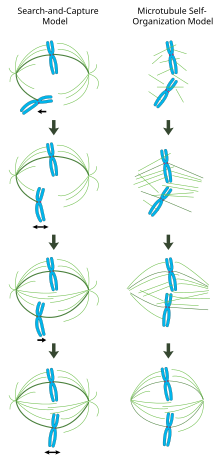
In a properly formed mitotic spindle, bi-oriented chromosomes are aligned along the equator of the cell with spindle microtubules oriented roughly perpendicular to the chromosomes, their plus-ends embedded in kinetochores and their minus-ends anchored at the cell poles. The precise orientation of this complex is required to ensure accurate chromosome segregation and to specify the cell division plane. However, it remains unclear how the spindle becomes organized. Two models predominate the field, which are synergistic and not mutually exclusive. In the search-and-capture model, the spindle is predominantly organized by the poleward separation of centrosomal microtubule organizing centers (MTOCs). Spindle microtubules emanate from centrosomes and 'seek' out kinetochores; when they bind a kinetochore they become stabilized and exert tension on the chromosomes. In an alternative self assembly model, microtubules undergo acentrosomal nucleation among the condensed chromosomes. Constrained by cellular dimensions, lateral associations with antiparallel microtubules via motor proteins, and end-on attachments to kinetochores, microtubules naturally adopt a spindle-like structure with chromosomes aligned along the cell equator.
Centrosome-mediated "search-and-capture" model
In this model, microtubules are nucleated at microtubule organizing centers and undergo rapid growth and catastrophe to 'search' the cytoplasm for kinetochores. Once they bind a kinetochore, they are stabilized and their dynamics are reduced. The newly mono-oriented chromosome oscillates in space near the pole to which it is attached until a microtubule from the opposite pole binds the sister kinetochore. This second attachment further stabilizes kinetochore attachment to the mitotic spindle. Gradually, the bi-oriented chromosome is pulled towards the center of the cell until microtubule tension is balanced on both sides of the
In this model, microtubule organizing centers are localized to the cell poles, their separation driven by microtubule polymerization and 'sliding' of antiparallel spindle microtubules with respect to one another at the spindle midzone mediated by bipolar, plus-end-directed kinesins.[19][20] Such sliding forces may account not only for spindle pole separation early in mitosis, but also spindle elongation during late anaphase.
Chromatin-mediated self-organization of the mitotic spindle
In contrast to the search-and-capture mechanism in which centrosomes largely dictate the organization of the mitotic spindle, this model proposes that microtubules are nucleated acentrosomally near chromosomes and spontaneously assemble into anti-parallel bundles and adopt a spindle-like structure.[21] Classic experiments by Heald and Karsenti show that functional mitotic spindles and nuclei form around DNA-coated beads incubated in Xenopus egg extracts and that bipolar arrays of microtubules are formed in the absence of centrosomes and kinetochores.[22] Indeed, it has also been shown that laser ablation of centrosomes in vertebrate cells inhibits neither spindle assembly nor chromosome segregation.[23] Under this scheme, the shape and size of the mitotic spindle are a function of the biophysical properties of the cross-linking motor proteins.[24]
Chromatin-mediated microtubule nucleation by the Ran GTP gradient
The guanine nucleotide exchange factor for the small GTPase
Regulation of spindle assembly
Spindle assembly is largely regulated by phosphorylation events catalyzed by mitotic kinases.
Cyclin dependent kinase complexes (CDKs) are activated by mitotic cyclins, whose translation increases during mitosis. CDK1 (also called CDC2) is considered the main mitotic kinase in mammalian cells and is activated by Cyclin B1.
Aurora kinases are required for proper spindle assembly and separation.
Mitotic chromosome structure
By the end of
While these dynamic rearrangements are vitally important to ensure accurate and high-fidelity segregation of the genome, our understanding of mitotic chromosome structure remains largely incomplete. A few specific molecular players have been identified, however: Topoisomerase II uses ATP hydrolysis to catalyze decatenation of DNA entanglements, promoting sister chromatid resolution.[33] Condensins are 5-subunit complexes that also use ATP-hydrolysis to promote chromosome condensation.[34] Experiments in Xenopus egg extracts have also implicated linker Histone H1 as an important regulator of mitotic chromosome compaction.[35]
Mitotic spindle assembly checkpoint
The completion of spindle formation is a crucial transition point in the cell cycle called the
Spindle apparatus orientation

Cell division orientation is of major importance for tissue architecture, cell fates and morphogenesis. Cells tend to divide along their long axis according to the so-called Hertwig rule. The axis of cell division is determined by the orientation of the spindle apparatus. Cells divide along the line connecting two centrosomes of the spindle apparatus. After formation, the spindle apparatus undergoes rotation inside the cell. The astral microtubules originating from centrosomes reach the cell membrane where they are pulled towards specific cortical clues. In vitro, the distribution of cortical clues is set up by the adhesive pattern.[38] In vivo polarity cues are determined by localization of Tricellular junctions localized at cell vertices.[39] The spatial distribution of cortical clues leads to the force field that determine final spindle apparatus orientation and the subsequent orientation of cell division.
See also
References
- PMID 18275887.
- S2CID 8145444.
- PMID 19889968.
- ISBN 0-8053-7171-0.
- S2CID 37305534.
- PMID 23415226.
- PMID 1970824.
- PMID 9585405.
- S2CID 11948950.
- PMID 12446741.
- PMID 20498706.
- PMID 16914514.
- S2CID 24977579.
- PMID 12388761.
- ^ PMID 21866268.
- PMID 11866534.
- ^ PMID 15548419.
- S2CID 10732643.
- PMID 19866774.
- PMID 9885249.
- PMID 19843451.
- S2CID 4238425.
- S2CID 9976687.
- PMID 17702580.
- PMID 20739938.
- PMID 22215983.
- ^ Fu J, Jiang Q, Zhang C (2010). "Coordination of Cell Cycle Events by Ran GTPase". Nature Education. 3 (9): 32.
- PMID 17715155.
- S2CID 22213611.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - ^ ISBN 978-0-9539181-2-6
- PMID 24529248.
- PMID 11395412.
- PMID 22855829.
- PMID 15967810.
- ISBN 0-7167-1007-2.
- PMID 16226453.
- S2CID 4391685.
- PMID 26886796.
External links
 Media related to Spindle apparatus at Wikimedia Commons
Media related to Spindle apparatus at Wikimedia Commons
