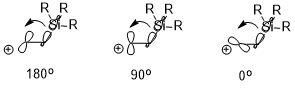Stereoelectronic effect
This article needs attention from an expert in Chemistry. The specific problem is: Numerous errors in the examples. (December 2019) |
In
Stereoelectronic effects present themselves in other well-known interactions. These include important phenomena such as the anomeric effect and hyperconjugation. It is important to note that stereoelectronic effects should not be misunderstood as a simple combination of steric effects and electronic effects.
Founded on a few general principles that govern how orbitals interact, the stereoelectronic effect, along with the steric effect,
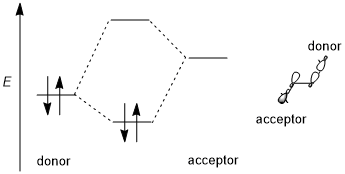
A stereoelectronic effect generally involves a stabilizing donor-acceptor (i.e., filled bonding-empty antibonding, 2-electron 2-orbital) interaction. The donor is usually a higher bonding or
Trend of different orbitals
Take the simplest CH2X–CH3 system as an example; the donor orbital is σ(C–H) orbital and the acceptor is σ*(C–X). When moving from fluorine to chlorine, then to bromine, the electronegativity of the halogen and the energy level of the σ*(C–X) orbitals decreases.[6] Consequently, the general trend of acceptors can be summarized as: π*(C=O)>σ*(C–Hal)>σ*(C–O)>σ*(C–N)>σ*(C–C), σ*(C–H). For donating orbitals, the nonbonding orbitals, or the lone pairs, are generally more effective than bonding orbitals due to the high energy levels. Also, different from acceptors, donor orbitals require less polarized bonds. Thus, the general trends for donor orbitals would be: n(N)>n(O)>σ(C–C), σ(C–H)>σ(C–N)>σ(C–O)>σ(C–S)>σ(C–Hal).[5]
Stereoelectronic effect can be directional in specific cases. The radius of
Influence on stability
If there is an
Influence on conformation
Gauche effect
One structural consequence of acyclic systems due to the stereoelectronic effect is the
This gauche effect and its impact on conformation are important in biochemistry. For example, in HIF-α subunit fragments containing (2S,4R)-4-hydroxyproline, the gauche interaction favors the conformer that can bind to the active site of pVHL.
Special effects of fluorine substituent
Stereoelectronic effects can have a significant influence in
Further studies illustrate that even for only one or two hydrogen atoms in a methyl group being replaced by a fluorine atom, the distortion in the structure can also be significant, with the [C(aryl)–C(aryl)–O–C(H2F)] dihedral angle in the energy minimized structure being around 24° and the [C(aryl)–C(aryl)–O–C(HF2)] dihedral angle 33°.[11]
Influence on reaction selectivity
Reductive cyclizations
Although the energy difference between coplanar anisole and its
The intermediate of the above reaction is the di-anion and the stereoelectronic effect that stabilizes this intermediate over the other one is the fact that the anionic charge at the para position could delocalize to the oxygen atom via orbital interaction: π(benzene) → σ*(O–CH3).[12]
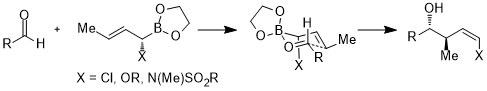
Hydrogenation
Even remote substituents on the
The relevant stereoelectronic interaction in the starting material is the nO → σ*(Ccarbonyl–Caryl) interaction. The electron-withdrawing substituent on the benzene ring depletes the electron density on the aromatic ring and thus makes the σ*(Ccarbonyl–Caryl(nitro)) orbital a better acceptor than σ*(Ccarbonyl–Caryl(methoxy)). These two stereoelectronic interactions use different lone pairs on the oxygen atom (the one antiperiplanar to the σ* in question for each), leading to lone pairs with different electron densities. In particular, the enhanced depletion of electron density from the lone pair antiperiplanar to the 4-nitrophenyl group leads to weakened ability for that lone pair to coordinate to boron. This in turn results in the lone pair antiperiplanar to the 4-methoxyphenyl binding preferentially to the catalyst, leading to well-defined facial selectivity. Under optimized conditions, the product is formed with excellent levels of enantioselectivity (95% ee).[13]

Influence on thermodynamics
Influence on equilibrium
The stereoelectronic effect influences the
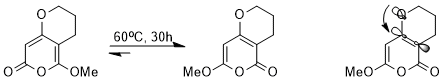
Despite very similar structures, one of the two isomers is strongly favored over the other because of a stereoelectronic effect. Since the σ*C-C orbital adjacent to the electron-withdrawing carbonyl group is lower in energy and is therefore a better acceptor than the σ*C-C orbital adjacent to the methoxy, the isomer in which the nO(σ) lone pair is able to donate into this lower-energy antibonding orbital will be stabilized (orbital interaction illustrated).[14]
Another example of the preference in the equilibrium within the area of pericyclic reaction is shown below. The stereoelectronic effect that affect the equilibrium is the interaction between the delocalized “banana bonds” and the empty p orbital on the boron atom.[15]
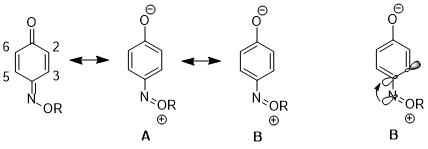
Influence on resonance structures
In another case, the stereoelectronic effect can result in an increased contribution of one resonance structure over another, which leads to further consequences in reactivity. For 1,4-benzoquinone monoxime, there are significant differences in the physical properties and reactivities between C2-C3 double bond and C5-C6 double bond. For instance, in the 1H NMR, 3J23 higher than 3J56.[16] The C2-C3 double bond also selectively undergoes Diels–Alder reaction with cyclopentadiene, despite the increased steric hindrance on that side of the molecule.[17] These data illustrate an increased contribution of resonance structure B over structure A. The authors argue that the donation from nN to σ*C4-C3 orbital lengthens the C4–C3 bond (C4 is the carbon bearing the nitrogen substituent), which reduces the p-p overlap between these two atoms. This in turn decreasing the relative importance of structure A which has a double bond between C4 and C3.[18]
Application in asymmetric Diels–Alder reactions
In the asymmetric Diels–Alder reactions, instead of using
The stereoelectronic effect affecting the outcome of the facial selectivity of the diene in the Diels–Alder reaction is the interaction between the σ(C(sp2)–CH3) (when σ(C(sp2)–X) is a better acceptor than a donor) or σ(C(sp2)–X) (when σ(C(sp2)–X) is a better donor than an acceptor) and the σ* orbital of the forming bond between the diene and the dienophile.[20]
If the two geminal substituents are both aromatic rings with different substituents tuning the electron density, the differentiation of the facial selectivity is also facile where the dienophile approaches the diene anti to the more electron-rich C–C bond where the stereoelectronic effect, in this case, is similar to the previous one.[21]
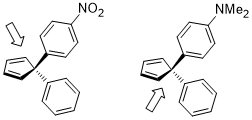
The ring opening of cyclobutene under heating conditions can have two products: inward and outward rotation.
The inward rotation transition state of the structure shown below is relatively favored for acceptor R substituents (e.g. NO2) but is especially disfavored by donor R substituents (e.g. NMe2).[22]
Stereoelectronic effect versus steric clash
Sometimes, stereoelectronic effects can win over extreme steric clash. In a similar cyclobutene ring-opening reaction, the trimethylsilyl group, which is very bulky, still favors the inward rotation. The stereoelectronic effect, which is the interaction shown above when the acceptor orbital is the σ*(Si–CH3), appears to be a more predominant factor in determining the reaction selectivity against the steric hindrance and even wins over the penalty of the disrupted conjugation system of the product due to steric clash.[23]
Furthermore, the acceptor orbitals are not limited to the antibonding orbitals of carbon-heteroatom bonds or the empty orbitals; in the following case, the acceptor orbital is the σ*(B–O) orbital. In the six-membered ring transition state, the stereoelectronic interaction is σ(C–X) → σ*(B–O).[24]
Stereoelectronic effects in biomolecules
Molecular recognition events mediated through orbital interactions are critical in a number of biological processes such as enzyme catalysis.[25] Stabilizing interactions between proteins and carbohydrates in glycosylated proteins also exemplify the role of stereoelectronic effects in biomolecules.[26]
References
- ^ a b Alabugin, I. V. Stereoelectronic Effects: the Bridge between Structure and Reactivity. John Wiley & Sons Ltd, Chichester, UK, 2016. http://eu.wiley.com/WileyCDA/WileyTitle/productCd-1118906349.html
- ISSN 0166-1280.
- ^ a b Evans, D. A. (2006). Chemistry 206 Lecture Notes. Cambridge, MA: Harvard University (Unpublished). pp. 1–2 (Lecture 1).
- OCLC 9412829.
- ^ ISBN 9780198558934.
- ^ PMID 11902907.
- ISBN 978-0387448978.
- ^ Hanack, M. (1965). Conformational Theory. New York and London: Academic Press.
- ISBN 1891389319.
- PMID 19295524.
- ^ S2CID 8905635.
- ^ PMID 23639080.
- ^ ISSN 0040-4039.
- ISSN 0040-4039.
- ISSN 0002-7863.
- .
- .
- PMID 11671465.
- .
- ^ PMID 10813872.
- .
- .
- PMID 15372640.
- .
- ISSN 0009-2665.
- PMID 33723379.