Sulfonate
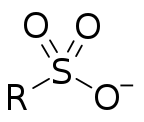
In
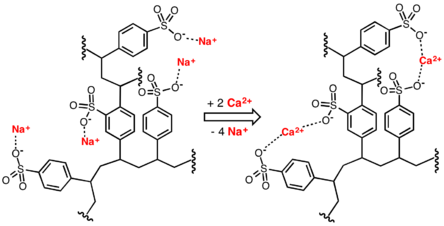
Sulfonate salts
A classic preparation of sulfonates is the Strecker sulfite alkylation, in which an alkali sulfite salt displaces a halide, typically in the presence of an iodine catalyst:[1]
An alternative is the condensation of a sulfonyl halide with an alcohol in pyridine:[2]
Sulfonic esters
Esters with the general formula R1SO2OR2 are called sulfonic esters. Individual members of the category are named analogously to
Sulfonic esters are used as reagents in organic synthesis, chiefly because the RSO3− group is a good
Sulfonates are commonly used to confer water solubility to protein crosslinkers such as N-hydroxysulfosuccinimide (Sulfo-NHS),
Sultones

Cyclic sulfonic esters are called sultones.
Tisocromide is an example of a sultone.
Examples
- Mesylate (methanesulfonate), CH3−SO−3
- Triflate (trifluoromethanesulfonate), CF3−SO−3
- Ethanesulfonate(esilate, esylate), CH3CH2−SO−3
- Tosylate(p-toluenesulfonate), p-CH3−C6H4−SO−3
- Benzenesulfonate (besylate), C6H5−SO−3
- Closilate (closylate, chlorobenzenesulfonate), Cl−C6H4−SO−3
- Camphorsulfonate(camsilate, camsylate), (C10H15O)−SO−3
- Pipsylate (p-iodobenzenesulfonate derivative), p-I−C6R4−SO−3, where R is any group.[5]
- Nosylate(o- or p-nitrobenzenesulfonate), o- or p-O2N−C6H4−SO−3
See also
References
- ISBN 978-0-387-44899-2.
- ISBN 0-471-95512-4
- PMID 22742473.
- PMID 5127991.


