Sulfoxide

In
Structure and bonding

Sulfoxides feature relatively short S–O distances. In DMSO, the S–O distance is 1.531 Å. The sulfur center is pyramidal; the sum of the angles at sulfur is about 306°.[3] Sulfoxides are generally represented with the structural formula R−S(=O)−R', where R and R' are organic groups. The bond between the
Chirality
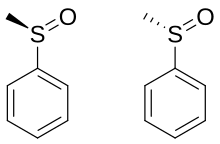
A
Preparation
Sulfoxides are typically prepared by
Symmetrical sulfoxides can be formed from a diorganylzinc compound and liquid sulfur dioxide.[11]
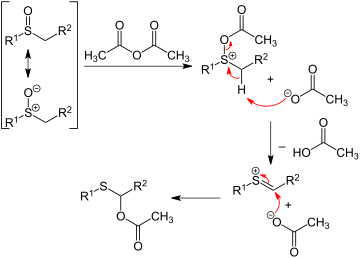
Aryl sulfoxides
In addition to the oxidation routes, di
- 2 ArH + SO2 → Ar2SO + H2O
Both aryl sulfinyl chlorides and diaryl sulfoxides can be also prepared from arenes through reaction with thionyl chloride in the presence of Lewis acid catalysts such as BiCl3, Bi(OTf)3, LiClO4, or NaClO4.[12][13]
Reactions
Deoxygenation and oxygenation
Sulfoxides undergo deoxygenation to give sulfides. Typically metal complexes are used to catalyze the reaction, using hydrosilanes as the stoichiometric reductant.[14] The deoxygenation of dimethylsulfoxide is catalyzed by DMSO reductase, a molybdoenzyme:[15]
- OSMe2 + 2 e− + 2 H+ → SMe2 + H2O
Acid-base reactions
The α-CH groups of alkyl sulfoxides are susceptible to deprotonation by strong bases, such as sodium hydride:[16]
- CH3S(O)CH3 + NaH → CH3S(O)CH2Na + H2
In the
Elimination reactions
Sulfoxide undergo thermal elimination via an Ei mechanism to yield vinyl alkenes and sulfenic acids.[17][18]
- CH3S(O)CH2CH2R → CH3SOH + CH2=CHR
The acids are powerful
The reverse reaction is possible.Coordination chemistry

Sulfoxides, especially DMSO, form coordination complexes with transition metals. Depending on the hard-soft properties of the metal, the sulfoxide binds through either the sulfur or the oxygen atom. The latter is particularly common.[22]
Applications and occurrence
DMSO is a widely used solvent.
The sulfoxide functional group occurs in several drugs. Notable is esomeprazole, the optically pure form of the proton-pump inhibitor omeprazole. Another commercially important sulfoxides include armodafinil.
Methionine sulfoxide forms from the amino acid methionine and its accumulation is associated with aging. The enzyme DMSO reductase catalyzes the interconversion of DMSO and dimethylsulfide.
Naturally-occurring chiral sulfoxides include alliin and ajoene.
Further reading
- Gama Á, Flores-López LZ, Aguirre G, Parra-Hake M, Hellberg LH, Somanathan R (2003). "Oxidation of sulfides to chiral sulfoxides using Schiff base-vanadium (IV) complexes". Arkivoc. 2003 (11): 4–15. .
References
- ISBN 9780470666357.
- .
- ..
- doi:10.1039/A700708F.
- S2CID 98211634.
- PMID 12964880.
- ISBN 978-3527306732.
- .
- ISBN 978-0471936237.
- .
- LCCN 52-12057.
- .
- S2CID 96348273.
- .
- S2CID 102494853.
- ^ Iwai I, Ide J (1988). "2,3-Diphenyl-1,3-Butadiene". Organic Syntheses; Collected Volumes, vol. 6, p. 531.
- doi:10.15227/orgsyn.070.0029.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 11749600.
- .
- ISBN 978-0-12-803581-8.
- .
- .