Synthon
This article may be too technical for most readers to understand. (September 2015) |
In
E. J. Corey.[1] He noted in 1988 that the "word synthon has now come to be used to mean synthetic building block rather than retrosynthetic fragmentation structures".[2]
It was noted in 1998[3] that the phrase did not feature very prominently in Corey's 1981 book The Logic of Chemical Synthesis,[4] as it was not included in the index. Because synthons are charged, when placed into a synthesis an uncharged form is found commercially instead of forming and using the potentially very unstable charged synthons.
Example
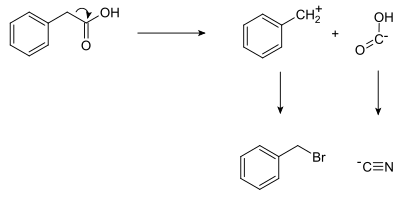
In planning the synthesis of
benzyl
synthon.
The synthesis of phenylacetic acid determined by retrosynthetic analysis is thus:
- Ph−CH2−Br + Na+[C≡N]− → Ph−CH2−C≡N + NaBr
- Ph−CH2−C≡N + 2 H2O → Ph−CH2−C(=O)−OH + NH3
where Ph stands for
phenyl
.
- C2 synthons - acetylene, acetaldehyde
- -C2H4OH synthon - ethylene oxide
- alkyl halides
- organolithiums, substituted acetylides
Alternative use in synthetic oligonucleotides
This term is also used in the field of gene synthesis—for example "40-base synthetic oligonucleotides are built into 500- to 800-bp synthons".[5]
Carbocationic synthons

Many retrosynthetic disconnections important for organic synthesis planning use carbocationic synthons.
carbonyl compounds such as ketones, aldehydes and carboxylic acid derivatives. An oxonium-type synthon was used in a disconnection en route[clarification needed] to the hops ether[clarification needed],[6] a key component of beer (see fig.1). In the forward direction, the researchers used an intramolecular aldol reaction catalyzed by titanium tetrachloride to form the tetrahydrofuran
ring of hops ether.
Another common disconnection that features carbocationic
Pictet-Spengler reaction has been used extensively for the synthesis of numerous indole and isoquinoline alkaloids.[7]
Celestolide (4-acetyl-6-t-butyl-1,1-dimethylindane, a component of musk perfume) can be synthesized using a benzyl anion alkylation with 3-chloro-2-methyl-1-propene as an intermediate step.[8]
The synthesis is fairly straightforward, and has been adapted for teaching purposes in an undergraduate laboratory.
References
- S2CID 73595158.
- .
- ISBN 0-85404-544-9.)
{{cite book}}: CS1 maint: multiple names: authors list (link - ISBN 0-471-11594-0.
- PMID 15496466.
- PMID 22148812.
- ISBN 0471264180.
- .