Titanium tetrachloride
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Titanium(IV) chloride
| |
| Other names
Titanium tetrachloride
Tetrachlorotitanium | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.028.584 |
| EC Number |
|
| MeSH | Titanium+tetrachloride |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 1838 |
CompTox Dashboard (EPA)
|
|
| |
SMILES
| |
| Properties | |
| TiCl4 | |
| Molar mass | 189.679 g/mol |
| Appearance | Colourless liquid |
| Odor | penetrating acid odor |
| Density | 1.726 g/cm3 |
| Melting point | −24.1 °C (−11.4 °F; 249.1 K) |
| Boiling point | 136.4 °C (277.5 °F; 409.5 K) |
| reacts (exothermic hydrolysis)[1] | |
| Solubility | soluble in dichloromethane,[2] toluene,[3] pentane[4] |
| Vapor pressure | 1.3 kPa (20 °C) |
| −54.0·10−6 cm3/mol | |
Refractive index (nD)
|
1.61 (10.5 °C) |
| Viscosity | 827 μPa s |
| Structure | |
| Tetragonal | |
| Tetrahedral | |
| 0 D | |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
355 J·mol−1·K−1[5] |
Std enthalpy of (ΔfH⦵298)formation |
−763 kJ·mol−1[5] |
| Hazards[6] | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Toxic, corrosive, reacts with water to release HCl |
| GHS labelling: | |
  
| |
| Danger | |
| H314, H317, H330, H335, H370, H372 | |
| P280, P301+P330+P331, P304+P340, P305+P351+P338, P308+P310 | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | MSDS |
| Related compounds | |
Other anions
|
Titanium(IV) iodide
|
Other cations
|
Hafnium(IV) chloride
Zirconium(IV) chloride |
Related compounds
|
Titanium(II) chloride Titanium(III) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Titanium tetrachloride is the inorganic compound with the formula TiCl4. It is an important intermediate in the production of titanium metal and the pigment titanium dioxide. TiCl4 is a volatile liquid. Upon contact with humid air, it forms thick clouds of titanium dioxide (TiO2) and hydrochloric acid, a reaction that was formerly exploited for use in smoke machines. It is sometimes referred to as "tickle" or "tickle 4", as a phonetic representation of the symbols of its molecular formula (TiCl4).[7][8]
Properties and structure
TiCl4 is a dense, colourless liquid, although crude samples may be yellow or even red-brown. It is one of the rare transition metal halides that is a liquid at room temperature, VCl4 being another example. This property reflects the fact that molecules of TiCl4 weakly self-associate. Most metal chlorides are polymers, wherein the chloride atoms bridge between the metals. Its melting point is similar to that of CCl4.[9][10]
Ti4+ has a "closed" electronic shell, with the same number of electrons as the noble gas
TiCl4 is soluble in
TiCl4L.Production
TiCl4 is produced by the chloride process, which involves the reduction of titanium oxide ores, typically ilmenite (FeTiO3), with carbon under flowing chlorine at 900 °C. Impurities are removed by distillation.[10]
- 2 FeTiO3 + 7 Cl2 + 6 C → 2 TiCl4 + 2 FeCl3 + 6 CO
The coproduction of
Applications
Production of titanium metal
The world's supply of titanium metal, about 250,000 tons per year, is made from TiCl4. The conversion involves the reduction of the tetrachloride with magnesium metal. This procedure is known as the Kroll process:[14]
- 2 Mg + TiCl4 → 2 MgCl2 + Ti
In the Hunter process, liquid sodium is the reducing agent instead of magnesium.[15]
Production of titanium dioxide
Around 90% of the TiCl4 production is used to make the pigment titanium dioxide (TiO2). The conversion involves hydrolysis of TiCl4, a process that forms hydrogen chloride:[14]
- TiCl4 + 2 H2O → TiO2 + 4 HCl
In some cases, TiCl4 is oxidised directly with oxygen:
- TiCl4 + O2 → TiO2 + 2 Cl2
Smoke screens
It has been used to produce smoke screens since it produces a heavy, white smoke that has little tendency to rise. "Tickle" was the standard means of producing on-set smoke effects for motion pictures, before being phased out in the 1980s due to concerns about hydrated HCl's effects on the respiratory system.[16]
Chemical reactions
Titanium tetrachloride is a versatile reagent that forms diverse derivatives including those illustrated below.[17]
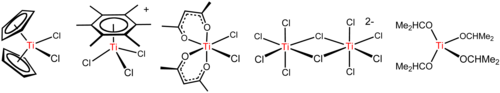
Alcoholysis and related reactions
A characteristic reaction of TiCl4 is its easy
Alcohols react with TiCl4 to give alkoxides with the formula [Ti(OR)4]n (R =
Organic amines react with TiCl4 to give complexes containing amido (R2N−-containing) and imido (RN2−-containing) complexes. With ammonia, titanium nitride is formed. An illustrative reaction is the synthesis of tetrakis(dimethylamido)titanium Ti(N(CH3)2)4, a yellow, benzene-soluble liquid:[19] This molecule is tetrahedral, with planar nitrogen centers.[20]
- 4 LiN(CH3)2 + TiCl4 → 4 LiCl + Ti(N(CH3)2)4
Complexes with simple ligands
TiCl4 is a
Redox
Reduction of TiCl4 with
Organometallic chemistry
The organometallic chemistry of titanium typically starts from TiCl4. An important reaction involves sodium cyclopentadienyl to give titanocene dichloride, TiCl2(C5H5)2. This compound and many of its derivatives are precursors to Ziegler–Natta catalysts. Tebbe's reagent, useful in organic chemistry, is an aluminium-containing derivative of titanocene that arises from the reaction of titanocene dichloride with trimethylaluminium. It is used for the "olefination" reactions.[17]
Reagent in organic synthesis
TiCl4 finds occasional use in
Toxicity and safety considerations
Hazards posed by titanium tetrachloride generally arise from its reaction with water that releases
References
- S2CID 93971747.
- ^ "titanium(IV) chloride, 1M soln. in dichloromethane". Alfa Aesar. Retrieved 7 March 2018.
- ^ "Titanium(IV) chloride solution 1.0 M in toluene". Sigma-Aldrich. Retrieved 7 March 2018.
- ^ Butts, Edward H De. "patent US3021349A".
- ^ ISBN 978-0-618-94690-7.
- ^ "Classifications - CL Inventory". echa.europa.eu.
- ^ [1] Archived 2013-02-17 at the Wayback Machine American Chemistry Council – "Titanium Tetrachloride: Stepping Stone to Amazing Technology"
- ^ "Archived copy". Archived from the original on 2014-03-19. Retrieved 2013-04-10.
{{cite web}}: CS1 maint: archived copy as title (link) Iowa State University – "Chemistry Material Safety Data Sheets" - ^ Earnshaw, A.; Greenwood, N. (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann.
- ^ ISBN 978-3-527-30673-2.
- .
- ^ .
- ISBN 978-0-470-13252-4.
- ^ ISBN 978-3-527-30673-2.
- ISBN 978-0-19-965145-0.
- ^ The Royal Navy at War (DVD). London: Imperial War Museum. 2005.
- ^ ISBN 0-387-15784-0
- )
- .
- .
- .
- ^ Mariappan Periasamy (2002): "New synthetic methods using the TiCl4-NR3 reagent system", Arkivoc, p. 151-166.
- ^ Gundersen, L.-L.; Rise, F.; Undheim, K. (2004). "Titanium(IV) chloride". In Paquette, L. (ed.). Encyclopedia of Reagents for Organic Synthesis. New York, NY: J. Wiley & Sons.
General reading
- Holleman, A. F.; Wiberg, E. (2001). Inorganic Chemistry. San Diego, CA: Academic Press. ISBN 978-0-12-352651-9.
- ISBN 978-0-08-037941-8.

