Indole

| |

| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
1H-Indole[1] | |
| Other names
2,3-Benzopyrrole, ketole,
1-benzazole | |
| Identifiers | |
3D model (
JSmol ) |
|
| 3DMet | |
| 107693 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.004.019 |
| EC Number |
|
| 3477 | |
| KEGG | |
PubChem CID
|
|
RTECS number
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H7N | |
| Molar mass | 117.151 g·mol−1 |
| Appearance | White solid |
| Odor | Feces or jasmine like |
| Density | 1.1747 g/cm3, solid |
| Melting point | 52 to 54 °C (126 to 129 °F; 325 to 327 K) |
| Boiling point | 253 to 254 °C (487 to 489 °F; 526 to 527 K) |
| 0.19 g/100 ml (20 °C) Soluble in hot water | |
| Acidity (pKa) | 16.2 (21.0 in DMSO) |
| Basicity (pKb) | 17.6 |
| -85.0·10−6 cm3/mol | |
| Structure | |
| Pna21 | |
| Planar | |
| 2.11 D in benzene | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Skin sensitising |
| GHS labelling: | |
 
| |
| Danger | |
| H302, H311 | |
| P264, P270, P280, P301+P312, P302+P352, P312, P322, P330, P361, P363, P405, P501 | |
| Flash point | 121 °C (250 °F; 394 K) |
| Safety data sheet (SDS) | [1] |
| Related compounds | |
Other cations
|
Indolium |
Related aromatic
compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Indole is an
General properties and occurrence
Indole is a
When indole is a
Indole undergoes
The name indole is a
, since indole was first isolated by treatment of the indigo dye with oleum.History

Indole chemistry began to develop with the study of the dye indigo. Indigo can be converted to isatin and then to oxindole. Then, in 1866, Adolf von Baeyer reduced oxindole to indole using zinc dust.[7] In 1869, he proposed a formula for indole.[8]
Certain indole derivatives were important dyestuffs until the end of the 19th century. In the 1930s, interest in indole intensified when it became known that the indole substituent is present in many important alkaloids, known as indole alkaloids (e.g., tryptophan and auxins), and it remains an active area of research today.[9]
Biosynthesis and function
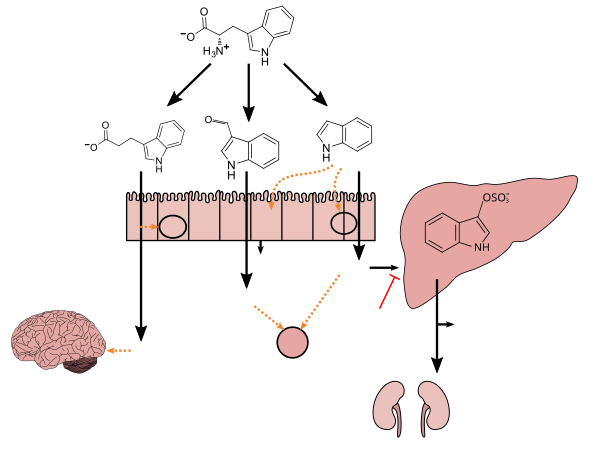
Indole is biosynthesized in the shikimate pathway via anthranilate.[2] It is an intermediate in the biosynthesis of tryptophan, where it stays inside the tryptophan synthase molecule between the removal of 3-phospho-glyceraldehyde and the condensation with serine. When indole is needed in the cell, it is usually produced from tryptophan by tryptophanase.[10]
As an
Tryptophan metabolism by
human gastrointestinal microbiota ( )activated charcoal), an intestinal sorbent that is taken by mouth, adsorbs indole, in turn decreasing the concentration of indoxyl sulfate in blood plasma.[12] |
As an
Detection methods
Common classical methods applied for the detection of extracellular and environmental indoles, are Salkowski, Kovács, Ehrlich’s reagent assays and HPLC.[16][17][18] For intracellular indole detection and measurement genetically encoded indole-responsive biosensor is applicable.[19]
Medical applications
Indoles and their derivatives are promising against
Synthetic routes
Indole and its derivatives can also be synthesized by a variety of methods.[25][26][27]
The main industrial routes start from
In general, reactions are conducted between 200 and 500 °C. Yields can be as high as 60%. Other precursors to indole include
Leimgruber–Batcho indole synthesis
The Leimgruber–Batcho indole synthesis is an efficient method of synthesizing indole and substituted indoles.[29] Originally disclosed in a patent in 1976, this method is high-yielding and can generate substituted indoles. This method is especially popular in the pharmaceutical industry, where many pharmaceutical drugs are made up of specifically substituted indoles.
Fischer indole synthesis

One of the oldest and most reliable methods for synthesizing substituted indoles is the
Other indole-forming reactions
- Bartoli indole synthesis
- Bischler–Möhlau indole synthesis
- Cadogan-Sundberg indole synthesis
- Fukuyama indole synthesis
- Gassman indole synthesis
- Hemetsberger indole synthesis
- Larock indole synthesis
- Madelung synthesis
- Nenitzescu indole synthesis
- Reissert indole synthesis
- Baeyer–Emmerling indole synthesis
- In the .
Chemical reactions of indole
Basicity
Unlike most amines, indole is not basic: just like pyrrole, the aromatic character of the ring means that the lone pair of electrons on the nitrogen atom is not available for protonation.[33] Strong acids such as hydrochloric acid can, however, protonate indole. Indole is primarily protonated at the C3, rather than N1, owing to the enamine-like reactivity of the portion of the molecule located outside of the benzene ring. The protonated form has a pKa of −3.6. The sensitivity of many indolic compounds (e.g., tryptamines) under acidic conditions is caused by this protonation.
Electrophilic substitution
The most reactive position on indole for
Since the pyrrolic ring is the most reactive portion of indole, electrophilic substitution of the carbocyclic (benzene) ring generally takes place only after N1, C2, and C3 are substituted. A noteworthy exception occurs when electrophilic substitution is carried out in conditions sufficiently acidic to exhaustively protonate C3. In this case, C5 is the most common site of electrophilic attack.[35]
Gramine, a useful synthetic intermediate, is produced via a Mannich reaction of indole with dimethylamine and formaldehyde. It is the precursor to indole-3-acetic acid and synthetic tryptophan.
N–H acidity and organometallic indole anion complexes
The N–H center has a pKa of 21 in
Carbon acidity and C2 lithiation
After the N–H proton, the hydrogen at C2 is the next most acidic proton on indole. Reaction of N-protected indoles with butyl lithium or lithium diisopropylamide results in lithiation exclusively at the C2 position. This strong nucleophile can then be used as such with other electrophiles.
Bergman and Venemalm developed a technique for lithiating the 2-position of unsubstituted indole,[37] as did Katritzky.[38]
Oxidation of indole
Due to the electron-rich nature of indole, it is easily oxidized. Simple oxidants such as N-bromosuccinimide will selectively oxidize indole 1 to oxindole (4 and 5).
Cycloadditions of indole
Only the C2–C3
Despite mediocre yields, intermolecular cycloadditions of indole derivatives have been well documented.
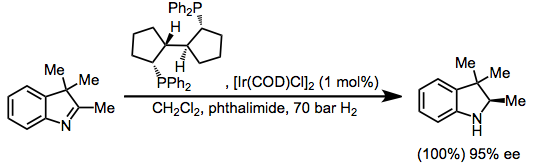
Hydrogenation
Indoles are susceptible to hydrogenation of the imine subunit.[45]
See also
- Indole-3-butyric acid
- Indole test
- Isoindole
- Isoindoline
- Skatole (3-methylindole)
References
- ISBN 978-0-85404-182-4.
- ^ ISBN 0-7167-4339-6.
- ^ Purves, Dale; Augustine, George J; Fitzpatrick, David; Katz, Lawrence C; LaMantia, Anthony-Samuel; McNamara, James O; Williams, S Mark. "Olfactory Perception in Humans". Olfactory Perception in Humans. Retrieved 20 October 2020.
- PMID 37901519.
- PMID 22450681.
- PMID 31640239.
- .
- .
- .
- ISBN 9780080536286.
- ^ PMID 20070374.
- ^ PMID 27102537.
Lactobacillus spp. convert tryptophan to indole-3-aldehyde (I3A) through unidentified enzymes [125]. Clostridium sporogenes convert tryptophan to IPA [6], likely via a tryptophan deaminase. ... IPA also potently scavenges hydroxyl radicals
Table 2: Microbial metabolites: their synthesis, mechanisms of action, and effects on health and disease
Figure 1: Molecular mechanisms of action of indole and its metabolites on host physiology and disease - PMID 19234110.
Production of IPA was shown to be completely dependent on the presence of gut microflora and could be established by colonization with the bacterium Clostridium sporogenes.
IPA metabolism diagram - ^ "3-Indolepropionic acid". Human Metabolome Database. University of Alberta. Retrieved 12 June 2018.
- S2CID 6630247.
[Indole-3-propionic acid (IPA)] has previously been identified in the plasma and cerebrospinal fluid of humans, but its functions are not known. ... In kinetic competition experiments using free radical-trapping agents, the capacity of IPA to scavenge hydroxyl radicals exceeded that of melatonin, an indoleamine considered to be the most potent naturally occurring scavenger of free radicals. In contrast with other antioxidants, IPA was not converted to reactive intermediates with pro-oxidant activity.
- PMID 188858.
- PMID 26386049.
- PMID 30050896.
- PMID 35563040.
- S2CID 218490655.
- S2CID 214695328.
- S2CID 198911553.
- S2CID 182950054.
- S2CID 219021072.
- doi:10.1039/a909834h.
- PMID 16011327.
- PMID 16836303.
- ISBN 978-3527306732.
- ^ "Indol NSP" (PDF).
- .
- .
- .
- ISBN 9781118681961.
- .
- .
- .
- .
- .
- PMID 12489950.
- .
- PMID 10891200.
- .
- ISBN 978-81-7736-278-7.
- PMID 12425597.
- ^ Zhu, G.; Zhang, X. Tetrahedron: Asymmetry 1998, 9, 2415.
General references
- Houlihan, W. J., ed. (1972). Indoles Part One. New York: Wiley Interscience.[ISBN missing]
- Sundberg, R. J. (1996). Indoles. San Diego: Academic Press. ISBN 978-0-12-676945-6.
- Joule, J. A.; Mills, K. (2000). Heterocyclic Chemistry. Oxford, UK: Blackwell Science. ISBN 978-0-632-05453-4.
- Joule, J. (2000). E. J., Thomas (ed.). Science of Synthesis. Vol. 10. Stuttgart: Thieme. p. 361. ISBN 978-3-13-112241-4.
- Schoenherr, H.; Leighton, J. L. (2012). "Direct and Highly Enantioselective Iso-Pictet-Spengler Reactions with α-Ketoamides: Access to Underexplored Indole Core Structures". Org. Lett. 14 (10): 2610–3. PMID 22540677.
External links
- Synthesis of indoles (overview of recent methods)
- Synthesis and properties of indoles at chemsynthesis.com