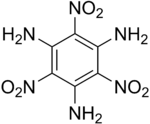
TATB
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
2,4,6-Trinitrobenzene-1,3,5-triamine | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.019.362 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H6N6O6 | |
| Molar mass | 258.15 g/mol |
| Appearance | Yellow or brown powdered crystals ( rhombohedral )
|
| Density | 1.93 g/cm3 |
| Melting point | 350 °C (662 °F; 623 K) |
| Explosive data | |
| Shock sensitivity | Insensitive |
| Friction sensitivity | Insensitive |
| Detonation velocity | 7350 m/s |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
TATB, triaminotrinitrobenzene or 2,4,6-triamino-1,3,5-trinitrobenzene is an
TATB is a very powerful explosive (somewhat less powerful than
TATB is normally used as the explosive ingredient in
Though it could theoretically be mixed with other explosive compounds in
Properties
At a pressed density of 1.80, TATB has a velocity of detonation of 7,350 meters per second.
TATB has a crystal density of 1.93 grams/cm3, though most forms currently in use have no higher density than 1.80 grams/cm3. TATB melts at 350 °C. The chemical formula for TATB is C6(NO2)3(NH2)3.
Pure TATB has a bright yellow color.
TATB has been found to remain stable at temperatures at least as high as 250 °C for prolonged periods of time.
Production
TATB is produced by
However, it is likely that the production of TATB will be switched over to a process involving the nitration and transamination of phloroglucinol, since this process is milder, cheaper, and reduces the amount of ammonium chloride salt produced in waste effluents (greener)[citation needed].
Still another process has been found for the production of TATB from materials that are surplus to military use.
See also
- FOX-7
- Plastic bonded explosive
- RE factor
Notes
- ^ Memorandum from Prospect, UK MOD position statement, 23 January 2006
- ^ David Albright (July 1994). "South Africa and the Affordable Bomb". Bulletin of the Atomic Scientists. p. 44.
- S2CID 54794595. UCRL-JC-122489.
- .
References
- Cooper, Paul W., Explosives Engineering, New York: Wiley-VCH, 1996. ISBN 0-471-18636-8
