Thiocarbamate
Appearance
organyl
(1) and S-organyl (2) thiocarbamatesIn
organosulfur compounds. As the prefix thio- suggests, they are sulfur analogues of carbamates. There are two isomeric forms of thiocarbamates: O-thiocarbamates, ROC(=S)NR2 (esters), and S-thiocarbamates, RSC(=O)NR2 (thioesters
).
Synthesis
Thiocarbamates can be synthesised by the reaction of water or alcohols upon thiocyanates (Riemschneider thiocarbamate synthesis):[1][2]
- RSCN + H2O → RSC(=O)NH2
- RSCN + R'OH → RSC(=O)NR'H
Similar reactions are seen between alcohols and thiocarbamoyl chlorides such as dimethylthiocarbamoyl chloride; as well as between thiols and cyanates.[2] The herbicide Cycloate is produced in this way:
- C6H11(C2H5)NCOCl + C2H5SH → C6H11(C2H5)NCOSC2H5 + HCl
Other related thiocarbamate herbicides include vernolate (C3H7)2NCOSC3H7 and triallate ((i−C3H7)2NCOSCH2CCl=CCl2.[3]
Salts of thiocarbamate arise by the reaction of amines with carbonyl sulfide:
- 2 R2NH + COS → [R2NH+2][R2N−COS−]
Reactions
In the
thiophenols
.

Occurrence
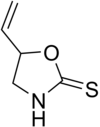
Goitrin is a cyclic thiocarbamate found in some vegetables.[5]
See also
- Dithiocarbamate
- Carbamate
- Tolnaftate, a thiocarbamate used as an antifungal agent
References
- ISBN 978-0-471-72091-1
- ^ .
- ISBN 3-527-30673-0.
- .
- PMID 2419242.
