Zinc stearate
| |
| Names | |
|---|---|
| IUPAC name
zinc octadecanoate
| |
| Other names
zinc distearate
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.008.321 |
| EC Number |
|
PubChem CID
|
|
RTECS number
|
|
| UNII | |
| UN number | 3077 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C36H70O4Zn | |
| Molar mass | 632.33 g·mol−1 |
| Appearance | soft, white powder |
| Odor | slight, characteristic[1] |
| Density | 1.095 g/cm3, solid |
| Melting point | 120 to 130 °C (248 to 266 °F; 393 to 403 K) |
| Boiling point | decomposes |
| insoluble | |
| Solubility in Ethanol | insoluble |
| Solubility in ether | insoluble |
| Solubility in benzene | slightly soluble |
| Hazards | |
| GHS labelling:[2] | |
 
| |
| Warning | |
| H335, H400, H413 | |
| P261, P271, P273, P304+P340, P312, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | 277 °C (531 °F; 550 K) |
| 420 °C (788 °F; 693 K) | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 15 mg/m3 (total) TWA 5 mg/m3 (resp)[1] |
REL (Recommended)
|
TWA 10 mg/m3 (total) TWA 5 mg/m3 (resp)[1] |
IDLH (Immediate danger) |
N.D.[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
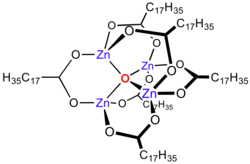
Zinc stearate is a "
Zinc carboxylates, e.g.
Applications
It is widely used as a release agent for the production of many kinds of objects: rubber, polyurethane, polyester processing system, powder metallurgy. These applications exploit its "non-stick" properties.[3] In cosmetics, zinc stearate is a lubricant and thickening agent used to improve texture.[4]
It is an "activator" for accelerated rubber sulfur vulcanization. As discovered in the early days of vulcanization, zinc has a beneficial effect on the reaction of the sulfur with the polyolefin. The stearate is a form of zinc that is highly soluble in the nonpolar medium of the polyolefins.
Being lipophilic, it functions as a
Niche uses
It is a component of some paints, imparting gloss. As a chief ingredient in "fanning powder", it is used by magicians performing card manipulation to decrease the friction between playing cards.
References
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0676". National Institute for Occupational Safety and Health (NIOSH).
- ^ "ZINC stearate". pubchem.ncbi.nlm.nih.gov.
- ^
- ^ "Zinc Stearate | Cosmetics Info". cosmeticsinfo.org. Archived from the original on 2013-09-06.

