2-Iodoxybenzoic acid

| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
1-Hydroxy-1λ5,2-benziodoxole-1,3-dione | |||
| Other names
1-Hydroxy-1λ3,2-benziodoxol-3(1H)-one 1-oxide
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 976364 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.157.592 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C7H5IO4 | |||
| Molar mass | 280.02 g/mol | ||
| Melting point | 233 °C (decomposes) | ||
| Hazards | |||
| GHS labelling: | |||
 
| |||
| Danger | |||
| H314, H315, H319, H335 | |||
| P260, P261, P264, P271, P280, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P332+P313, P337+P313, P362, P363, P403+P233, P405, P501 | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
2-Iodoxybenzoic acid (IBX) is an
Preparation
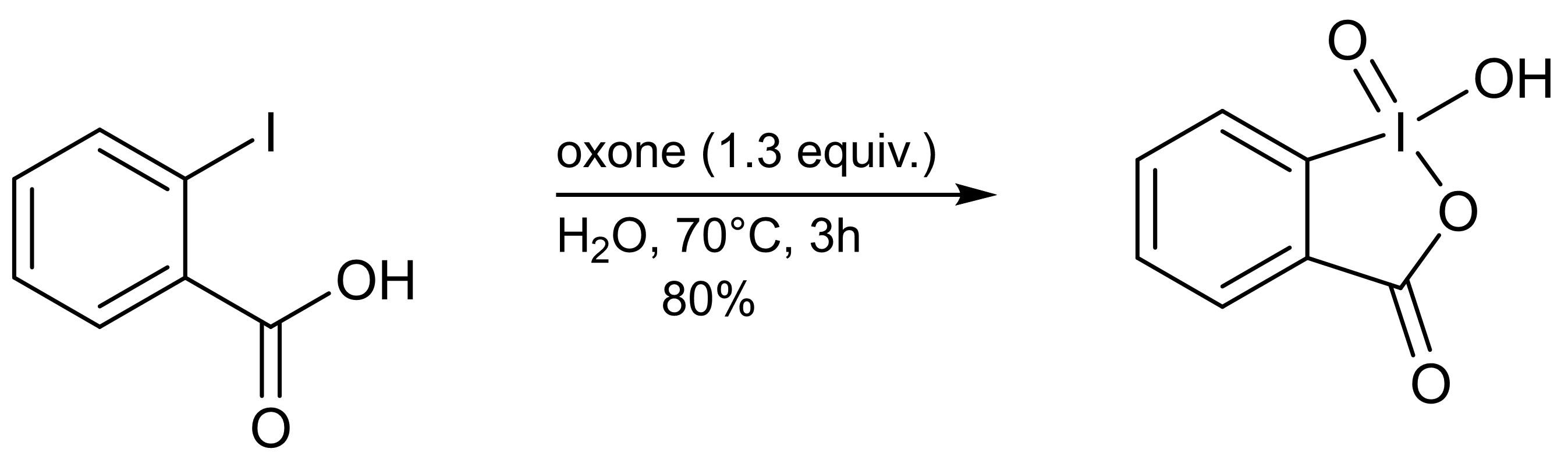
IBX can be prepared in a single step by adding an excess of
Reaction mechanism
The
IBX exists as two
Scope
IBX is also available as silica gel or polystyrene bound IBX. In many applications, IBX is replaced by Dess–Martin periodinane which is more soluble in common organic solvents. A sample reaction is an IBX oxidation used in the total synthesis of eicosanoid:[5] More and Finney[6] and Van Arman[7] have demonstrated that common organic solvents are suitable for many IBX oxidations, despite its low solubility, and in fact may simplify product purification.
- chemical yield(Mohapatra, 2005)
In 2001,
Oxidative cleavage
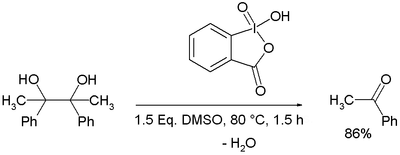
IBX is notable for oxidizing vicinal diols (or glycols) to diketones without cleavage of the carbon-carbon bond,[9] but oxidative cleavage of glycols to two aldehydes or ketones can occur when modified conditions are used (elevated temperatures or trifluoroacetic acid solvent).[10]
The reaction mechanism for this glycol cleavage is based on initial formation of an adduct between 10-I-4 IBX and DMSO to a 12-I-5 intermediate 3 in which DMSO acts as a leaving group for incoming alcohol 4 to intermediate 5. One equivalent of water is split off forming 12-I-5 spirobicyclic periodinane 6 setting the stage for fragmentation to 7. With hydroxyl alpha protons present, oxidation to the acyloin competes. Trifluoroacetic acid is found to facilitate the overall reaction.
α-Hydroxylations
Kirsch and co-workers were able to hydroxylate keto compounds with IBX in α-position under mild conditions.[11] This method could be extended to β-keto esters.[12]
Oxidation of β-hydroxyketones to β-diketones
Bartlett and Beaudry discovered that IBX is a valuable reagent for the transformation of β-hydroxyketones to β-diketones. IBX provides yields superior to both the Swern and Dess–Martin oxidation protocols.[13]
References
- ^ Boeckman, R. K. Jr.; Shao, P.; Mullins, J. J. (2000). "Dess–Martin periodinane: 1,1,1-Triacetoxy-1,1-dihydro-1,2-benziodoxol-3(1H)-one" (PDF). Organic Syntheses. 77: 141; Collected Volumes, vol. 10, p. 696.
- ^ .
- PMID 16218584.
- PMID 16566050.
- .
- PMID 12182609.
- .
- PMID 11878978.
- .
- PMID 17315062.
- PMID 16292876.
- PMID 19760735.
- PMID 22023037.