Dimethyl sulfoxide
| |||
| |||
 A sample of dimethyl sulfoxide
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
(Methanesulfinyl)methane | |||
| Systematic IUPAC name
(Methanesulfinyl)methane (substitutive) Dimethyl(oxido)sulfur (additive) | |||
| Other names
Methylsulfinylmethane
Methyl sulfoxide (2:1), Dermasorb[1] | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| Abbreviations | DMSO, Me2SO | ||
| 506008 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
ECHA InfoCard
|
100.000.604 | ||
| EC Number |
| ||
| 1556 | |||
| KEGG | |||
| MeSH | Dimethyl+sulfoxide | ||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C2H6OS | |||
| Molar mass | 78.13 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Density | 1.1004 g⋅cm−3 | ||
| Melting point | 19 °C (66 °F; 292 K) | ||
| Boiling point | 189 °C (372 °F; 462 K) | ||
| Miscible | |||
| Solubility in Diethyl ether | Not soluble | ||
| Vapor pressure | 0.556 millibars or 0.0556 kPa at 20 °C[2] | ||
| Acidity (pKa) | 35[3] | ||
Refractive index (nD)
|
1.479 εr = 48
| ||
| Viscosity | 1.996 cP at 20 °C | ||
| Structure | |||
| Cs | |||
| Trigonal pyramidal | |||
| 3.96 D | |||
| Pharmacology | |||
| G04BX13 (WHO) M02AX03 (WHO) | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Irritant | ||
| NFPA 704 (fire diamond) | |||
| Flash point | 89 °C (192 °F; 362 K) | ||
| Safety data sheet (SDS) | Oxford MSDS | ||
| Related compounds | |||
Related sulfoxides
|
Diethyl sulfoxide | ||
Related compounds
|
|||
| Supplementary data page | |||
| Dimethyl sulfoxide (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Dimethyl sulfoxide (DMSO) is an
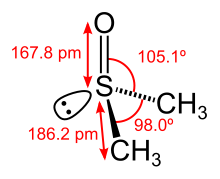
In terms of chemical structure, the molecule has idealized Cs symmetry. It has a trigonal pyramidal molecular geometry consistent with other three-coordinate S(IV) compounds,[6] with a nonbonded electron pair on the approximately tetrahedral sulfur atom.
Synthesis and production
Dimethyl sulfoxide was first synthesized in 1866 by the Russian scientist
Reactions
Reactions with electrophiles
The sulfur center in DMSO is
- (CH3)2SO + CH3I → [(CH3)3SO]I
This salt can be
:- [(CH3)3SO]I + NaH → (CH3)2S(CH2)O + NaI + H2
Acidity
The methyl groups of DMSO are only weakly acidic, with a pKa = 35. For this reason, the basicities of many weakly basic organic compounds have been examined in this solvent.
Deprotonation of DMSO requires strong bases like
Oxidant
In organic synthesis, DMSO is used as a mild oxidant.[10] It forms the basis of several selective sulfonium-based oxidation reactions including the Pfitzner–Moffatt oxidation, Corey–Kim oxidation and the Swern oxidation.[11] The Kornblum oxidation is conceptually similar. These all involve formation of an intermediate sulfonium species (R2S+X where X is a heteroatom)
Ligand and Lewis base
Related to its ability to dissolve many salts, DMSO is a common
In carbon tetrachloride solutions DMSO functions as a Lewis base with a variety of Lewis acids such as I2, phenols, trimethyltin chloride, metalloporphyrins, and the dimer Rh2Cl2(CO)4. The donor properties are discussed in the ECW model. The relative donor strength of DMSO toward a series of acids, versus other Lewis bases, can be illustrated by C-B plots.[13][14]
Applications
Solvent

DMSO is a
Because of its high boiling point, 189 °C (372 °F), DMSO evaporates slowly at normal atmospheric pressure. Samples dissolved in DMSO cannot be as easily recovered compared to other solvents, as it is very difficult to remove all traces of DMSO by conventional
In its
DMSO is used to dissolve test compounds in
DMSO is used as a vehicle in
DMSO is finding increased use in manufacturing processes to produce microelectronic devices.[24] It is widely used to strip photoresist in TFT-LCD 'flat panel' displays and advanced packaging applications (such as wafer-level packaging / solder bump patterning). DMSO is an effective paint stripper, being safer than many of the others such as nitromethane and dichloromethane.
Biology
DMSO is used in
DMSO in a PCR is applicable for supercoiled plasmids (to relax before amplification) or DNA templates with high GC-content (to decrease thermostability). For example, 10% final concentration of DMSO in the PCR mixture with Phusion decreases primer annealing temperature (i.e. primer melting temperature) by 5.5–6.0 °C (9.9–10.8 °F).[26]
It is well known as a reversible cell cycle arrester at phase G1 of human lymphoid cells.[27]
DMSO may also be used as a cryoprotectant, added to cell media to reduce ice formation and thereby prevent cell death during the freezing process.[28] Approximately 10% may be used with a slow-freeze method, and the cells may be frozen at −80 °C (−112 °F) or stored in liquid nitrogen safely.
In cell culture, DMSO is used to induce
Medicine
Use of DMSO in medicine dates from around 1963, when an
DMSO has been examined for the treatment of numerous conditions and ailments, but the U.S.
In
In
DMSO is metabolized by
Alternative medicine
DMSO is marketed as an alternative medicine. Its popularity as an alternative cure is stated to stem from a 60 Minutes documentary in 1980 featuring an early proponent.[33] However, DMSO is an ingredient in some products listed by the U.S. FDA as fake cancer cures[34] and the FDA has had a running battle with distributors.[33] One such distributor is Mildred Miller, who promoted DMSO for a variety of disorders and was consequently convicted of Medicare fraud.[33]
The use of DMSO as an alternative treatment for cancer is of particular concern, as it has been shown to interfere with a variety of chemotherapy drugs, including cisplatin, carboplatin, and oxaliplatin.[35] There is insufficient evidence to support the hypothesis that DMSO has any effect,[36] and most sources agree that its history of side effects when tested warrants caution when using it as a dietary supplement, for which it is marketed heavily with the usual disclaimer.
Veterinary medicine
DMSO is commonly used in veterinary medicine as a liniment for horses, alone or in combination with other ingredients. In the latter case, often, the intended function of the DMSO is as a solvent, to carry the other ingredients across the skin. Also in horses, DMSO is used intravenously, again alone or in combination with other drugs. It is used alone for the treatment of increased intracranial pressure and/or cerebral edema in horses.[citation needed]
Taste
The perceived garlic taste upon skin contact with DMSO may be due to
Safety
Toxicity
DMSO is a non-toxic solvent with a median lethal dose higher than ethanol (DMSO: LD50, oral, rat, 14,500 mg/kg;[38][39] ethanol: LD50, oral, rat, 7,060 mg/kg[40]).
DMSO can cause contaminants, toxins, and medicines to be absorbed through the skin, which may cause unexpected effects. DMSO is thought to increase the effects of blood thinners, steroids, heart medicines, sedatives, and other drugs. In some cases this could be harmful or dangerous.[41]
Because DMSO easily penetrates the
Regulation
In Australia, it is listed as a Schedule 4 (S4) Drug, and a company has been prosecuted for adding it to products as a preservative.[44]
Clinical safety
Early clinical trials with DMSO were stopped because of questions about its safety, especially its ability to harm the eye. The most commonly reported side effects include headaches and burning and itching on contact with the skin. Strong allergic reactions have been reported.[full citation needed]
On September 9, 1965, The Wall Street Journal reported that a manufacturer of the chemical warned that the death of an Irish woman after undergoing DMSO treatment for a sprained wrist may have been due to the treatment, although no autopsy was done, nor was a causal relationship established.[45] Clinical research using DMSO was halted and did not begin again until the National Academy of Sciences (NAS) published findings in favor of DMSO in 1972.[46] In 1978, the US FDA approved DMSO for treating interstitial cystitis. In 1980, the US Congress held hearings on claims that the FDA was slow in approving DMSO for other medical uses. In 2007, the US FDA granted "fast track" designation on clinical studies of DMSO's use in reducing brain tissue swelling following traumatic brain injury.[46]
DMSO exposure to developing mouse brains can produce brain degeneration. This
Odor problem
DMSO disposed into sewers can cause odor problems in municipal effluents: waste water bacteria transform DMSO under hypoxic (anoxic) conditions into dimethyl sulfide (DMS) that has a strong disagreeable odor, similar to rotten cabbage.[48] However, chemically pure DMSO is odorless because of the lack of C-S-C (sulfide) and C-S-H (mercaptan) linkages. Deodorization of DMSO is achieved by removing the odorous impurities it contains.[49]
Explosion hazard
Dimethyl sulfoxide can produce an explosive reaction when exposed to acyl chlorides; at a low temperature, this reaction produces the oxidant for Swern oxidation.
DMSO can decompose at the boiling temperature of 189 °C at normal pressure, possibly leading to an explosion. The decomposition is catalyzed by acids and bases and therefore can be relevant at even lower temperatures. A strong to explosive reaction also takes place in combination with halogen compounds, metal nitrides, metal perchlorates, sodium hydride, periodic acid and fluorinating agents.[50]
See also
- Varying oxidation of sulfur
- Dimethyl sulfide (DMS), the corresponding sulfide, also produced by marine phytoplankton and emitted to the oceanic atmosphere where it is oxidized to DMSO, SO2 and sulfate
- Dimethyl sulfone, commonly known as methylsulfonylmethane (MSM), a related chemical often marketed as a dietary supplement
- Related compounds with methyl on oxygen
- Dimethyl sulfite, the corresponding sulfite
- mutagenic alkylatingcompound
- Methyl methanesulfonate, another methylating agent
- Gloria Ramirez, also known as the "Toxic Woman"
References
- ^ DMSO (medication)
- ^ "Dimethyl Sulfoxide (DMSO) -- Technical". Atofina Chemicals, inc. Retrieved 26 May 2007.
- .
- ^ "Dimethyl sulfoxide". pubchem.ncbi.nlm.nih.gov.
- ISBN 978-1-57439-110-7.
- .
- ^ von Demselben (1867). "Ueber die Einwirkung von Saltpetersäure auf Schwefelmethyl und Schwefeläthyl" [On the effect of nitric acid on methyl sulfide and ethyl sulfide]. In Erlenmeyer, E.; Rieckher, T.; Volhard, J.; Liebig, J.; Wöhler, F. (eds.). Annalen der Pharmacie (in German). Meyer ; Winter. p. 148.
- ^ Gergel, Max G. (March 1977). Excuse me sir, would you like to buy a kilo of isopropyl bromide?. Pierce Chemical. p. 145.
- ISBN 3527306730
- PMID 6042131.
- .
- .
- OCLC 428031803.
- doi:10.1021/ed054p612. The plots shown in this paper used older parameters. Improved E&C parameters are listed in ECW model.
- ^ "DMSO". exactantigen.com. Archived from the original on 2009-10-05. Retrieved 2009-10-02.
- ^
Bordwell FG (1988). "Equilibrium acidities in dimethyl sulfoxide solution". S2CID 26624076.
- ^ "Bordwell pKa Table (Acidity in DMSO)". Archived from the original on 9 October 2008. Retrieved 23 April 2019.
- ^ S2CID 219590658.
- ^ PMID 17099243.
- ^ PMID 16472214.
- PMID 22216122.
- ^ a b Kelava T, Cavar I (Nov 2011). "Biological actions of drug solvents". Periodicum Biologorum. 113 (3): 311–320.
- ^ PMID 20962895.
- S2CID 137979405.
- ^
Chakrabarti R, Schutt CE (August 2001). "The enhancement of PCR amplification by low molecular-weight sulfones". PMID 11675022.
- ^ "Guidelines for PCR Optimization with Phusion High-Fidelity DNA Polymerase".
- ^
Sawai M, Takase K, Teraoka H, Tsukada K (1990). "Reversible G1 arrest in the cell cycle of human lymphoid cell lines by dimethyl sulfoxide". Exp. Cell Res. 187 (1): 4–10. PMID 2298260.
- ^ Pegg, DE (2007). "Principles of Cryopreservation". In Day JG, Stacey GN (eds.). Cryopreservation and Freeze-Drying Protocols. Methods in Molecular Biology. Vol. 368. Humana Press. pp. 39–57. )
- ISBN 978-0-7923-7195-3. Retrieved 2011-08-07.
- PMID 26491374.
- ^ "Import Alert 62-06". www.accessdata.fda.gov. Archived from the original on 2017-02-04. Retrieved 2017-03-05.
- ^
Shirley SW, Stewart BH, Mirelman S (March 1978). "Dimethyl Sulfoxide in Treatment of Inflammatory Genitourinary Disorders". PMID 636125.
- ^ a b c Jarvis WT (24 November 2001). "DMSO". National Council Against Health Fraud. Retrieved 19 July 2022.
- ^ "187 Fake Cancer "Cures" Consumers Should Avoid". FDA. Archived from the original on 23 July 2017.
- PMID 24812268.
- ^ Saling, Joseph (20 June 2022). "DMSO: Uses and Risks". WebMD. Retrieved 19 July 2022.
- PMID 24205061.
- ^ "Safety Data Sheet: Dimethyl Sulfoxide (DMSO)" (PDF). Gaylord Chemical Company, L.L.C. 21 July 2016. Archived (PDF) from the original on 13 February 2019.
- ^ "Material Safety Data Sheet: Dimethyl Sulfoxide". ScienceLab.com. 21 May 2013. Archived from the original on 19 September 2018.
- ^ "Material Safety Data Sheet: Ethyl alcohol 200 Proof". ScienceLab.com. 21 May 2013. Archived from the original on 19 September 2018.
- ^ "DMSO". American Cancer Society. Archived from the original on 27 July 2010.
- ^ Rubber Chemical Resistance Chart
- ^ "Chemical hygiene plan" (PDF). Cornell University. October 1999. Retrieved 2010-04-12.
- ^ "Brisbane drug company convicted of counterfeiting". Commonwealth of Australia: Department of Health and Ageing. 23 April 2003. Archived from the original on 2012-03-21.
- ^ Carley W (September 9, 1965). "DMSO may have caused death of woman, makers of 'Wonder' drug warn doctors". The Wall Street Journal. New York City.
- ^ a b https://www.fda.gov/ForIndustry/ImportProgram/ImportAlerts/ucm162294.htm [dead link]
- PMID 19100327.
- PMID 16433352.
- ^ US application 2009005601A1, George Kvakovszky; David Villarrubia II & Scott Stevenson et al., "Process for preparing low malodorous dimethyl sulfoxide", published 2009, assigned to Gaylord Chemical Company LLC
- ISBN 3-609-48040-8





