4-Hydroxycoumarins

4-Hydroxycoumarins are a class of
The primary mechanism of the 4-hydroxycoumarin drugs is the inhibition of vitamin K epoxide reductase. These compounds are not direct antagonists (in the pharmaceutical sense) of vitamin K, but rather act to deplete reduced vitamin K in tissues. For this reason vitamin K antagonizes their effect, and this has led to the loose terminology of "vitamin K antagonist".
Origin
Although 4-hydroxycoumarin itself is not an anticoagulant, it is an important fungal metabolite from the precursor
Effects
The synthetic drugs in the 4-hydroxycoumarin class are all noted primarily for their use as

Drugs and poisons in the class
The simplest synthetic molecule in the 4-hydroxycoumarin class is
Pharmaceutical examples of 4-hydroxycoumarin pharmaceuticals include:
Compounds in this class have also been used as pesticides, specifically rodenticides. They act by causing the affected animal to hemorrhage, causing it to seek water, and thus leave dwellings to die outdoors.
The second-generation vitamin K antagonist agents, used only in this fashion as poisons (because their duration of action is too long to be used as pharmaceuticals) include:
Structures

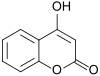

Coumarin
This molecule does not affect coagulationtobacco additivecoumarin.Dicumarol
This molecule was the first discovered 4-hydroxycoumarin anticoagulant. It is a dimer type structure connected at the 3 ring position.


Phenprocoumon
(anticoagulant)Warfarin
Most commonly used anticoagulant pharmaceuticalAcenocoumarol
(anticoagulant)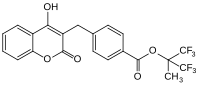
Tecarfarin (experimental anticoagulant)


Brodifacoum
This molecule is a second-generation anticoagulant with a large 3-position substituent which causes it to be retained in fatty tissues for longer times than first-generation compounds and pharmaceuticals. (rodenticide)Bromadiolone
(rodenticide)

Coumatetralyl
(rodenticide)Difenacoum
(rodenticide)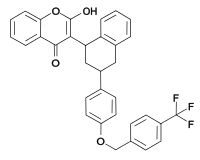
Flocoumafen
(rodenticide)
See also
References
- ^ Bye, A., King, H. K., 1970. The biosynthesis of 4-hydroxycoumarin and dicoumarol by Aspergillus fumigatus Fresenius. Biochemical Journal 117, 237-245.
External links
- 4-hydroxycoumarins at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Synthesis












