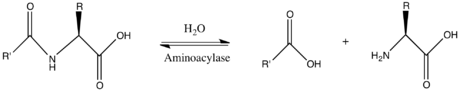
Aminoacylase
| aminoacylase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
In
- L-amino acid
Thus, the two
L-amino acid
.
This
urea cycle and metabolism of amino groups
.
Enzyme structure

As of late 2007, two
binding domains.[1][2] It is this dimerization that allows catalysis to occur, since aminoacylase's active site lies between its two Zinc binding domains.[1]
Bound
quaternary structure
.
Enzyme mechanism

Aminoacylase is a

The nucleophilic attack by water is the rate-limiting step of aminoacylase's
nucleophilic attack is reversible while the subsequent steps are fast and irreversible.[6] This reaction sequence is an example of Michaelis–Menten kinetics, allowing one to determine KM, Kcat, Vmax, turnover number, and substrate specificity through classic Michaelis-Menten enzyme experiments.[6] The second and third forward steps cause the formation and release of the reaction's products.[6]
Biological function

Aminoacylases are expressed in the
L-amino acids
and aid in urea cycle regulation.
energy
.
Aminoacylase is involved in the
molecules to the urea cycle.[7] The urea cycle gets rid of excess ammonia (NH4+) in the body, a process that must be up-regulated during times of increased protein catabolism, as amino acid breakdown produces large amounts of NH4+.[7] When amino acid catabolism increases, N-Acetylglutamate synthase is up-regulated, producing more N-acetyl-L-glutamate, which up-regulates carbamoyl phosphate synthetase and allows it to dispose of the excess NH4+ from catabolism.[7]
Aminoacylase is up-regulated during times of nutrient deficit or starvation, causing N-acetyl-L-glutamate breakdown, which down-regulates carbamoyl phosphate synthetase and the rest of the urea cycle. This response is evolutionarily advantageous, since a nutrient deficit means there isn't as much NH4+ that needs to be disposed of and since the body wants to salvage as many amino acids as it can.[7]
Disease relevance
autism .[16] Patients with A1D often start expressing symptoms shortly after birth but seem to recover fully in the next few years.[13][14][15]
aminoacylase 1 cannot.[17]
Industrial relevance
Aminoacylases have been used for the production of L-
amino acids in an enantiomerically
specific way.
Evolution
Many
enzymes evolved from a common ancestral protein, retaining function but diverging in structure over time.[1][4]
References
- ^ PMID 12933810.
- PMID 13061423.
- ^ PMID 18341290.
- ^ PMID 1284246.
- ^ PMID 15581567.
- ^ PMID 5160398.
- ^ ISBN 978-1-4292-2936-4.
- ^ PMID 21414403.
- S2CID 24017306.
- PMID 2542383.
- ^ EntrezGene 95
- PMID 1707030.
- ^ PMID 16465618.
- ^ S2CID 43376960.
- ^ PMID 16274666.
- S2CID 13374954.
- PMID 12824065.
- ^ ISBN 978-0-470-05458-1.
- PMID 14927637.
- PMID 8821711.
- PMID 16244442.
- PMID 8357837.