Barium ferrite
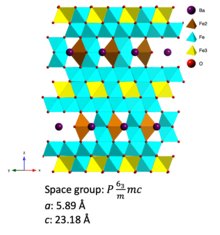 Barium ferrite crystal structure
| |
| Identifiers | |
|---|---|
3D model (
JSmol ) |
|
ECHA InfoCard
|
100.031.782 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| BaFe12O19 | |
| Molar mass | 1111.448 g·mol−1 |
| Appearance | black solid |
| Density | 5.28 g/cm3 |
| Melting point | 1,316 °C (2,401 °F; 1,589 K) |
| insoluble | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Barium
BaFe is described as Ba2+
Fe3+
12O2−
19. The Fe3+
centers are
Barium ferrite is a highly
Chemical structure
The Fe3+
centers, with a
A related family of industrially useful "hexagonal ferrites" are known, also containing
ions. Formulas for these species include BaFe
12O
19, BaFe
15O
23, and BaFe
18O
27.[6]
A one-step hydrothermal process can be used to form crystals of barium ferrite, by mixing
Properties
Barium ferrite has been considered for long term data storage. The material has proven to be resistant to a number of different environmental stresses, including humidity and corrosion. Because ferrites are already oxidized it can not be oxidized any further. This is one reason ferrites are so resistant to corrosion.[9] Barium ferrite also proved to be resistant to thermal demagnetization, another issue common with long-term storage.[4] The Curie temperature is typically around 450 °C (723K).
When barium ferrite magnets increase in temperature, their high intrinsic coercivity improves, this is what makes it more resistant to thermal demagnetization. Ferrite magnets are the only type of magnets that become substantially more resistant to demagnetization as temperature increases. This characteristic of barium ferrite makes it a popular choice in motor and generator designs and also in loudspeaker applications. Ferrite magnets can be used in temperatures up to 300 °C, which makes it a perfect to be used in the applications mentioned above. Ferrite magnets are extremely good insulators and don't allow any electrical current to flow through them and they are brittle which shows their ceramic characteristics. Ferrite magnets also have good machining properties, which allows for the material to be cut in many shapes and sizes.[10]
Chemical properties
Barium ferrites are robust ceramics that are generally stable to moisture and corrosion-resistant.[9] Ba‑Fe ferrite is an oxide, so it does not break down due to oxidation as much as a metal alloy might; giving Ba‑Fe a much greater life expectancy.[4]
Mechanical properties
Barium ferrite completely out classes MP, mostly because Ba‑Fe is already in its oxidized state and so is not restricted in its size by a protective coating. Also due to its hexagonal pattern it is easier to organize compared to the unorganized rod like MP. Another factor is the difference in the size of the particles, in MP the size ranges from 40 to 100 nm while the Ba‑Fe is only 20 nm. So the smallest MP particle is still double the size of the BaFe particles.[11]
Applications

Barium ferrite is used in applications such as recording media, permanent magnets, and magnetic stripe cards (credit cards, hotel keys, ID cards). Due to the stability of the material, it is able to be greatly reduced in size, making the packing density much greater. Earlier media devices utilized doped acicular oxide materials to yield the coercivity values necessary to record. In recent decades, barium ferrite has replaced acicular oxides; without any dopants, the acicular oxides produce very low coercivity values, making the material very magnetically soft, while barium ferrite's higher coercivity levels make the material magnetically hard and thus a superior choice for recording material applications.
Magnetic Stripes
ID cards using barium ferrite are made with a magnetic fingerprint that identifies them, allowing readers to self-calibrate.[12]
Speaker magnets
Barium ferrite is a common material for speaker magnets. The materials can be formed into almost any shape and size using a process called sintering, whereby powdered barium ferrite is pressed into a mold, and then heated until it fuses together. The barium ferrite turns into a solid block while still retaining its magnetic properties. The magnets have an excellent resistance to demagnetization, allowing them to still be useful in speaker units over a long period of time.[13]
Tape Data Storage Media
Barium ferrite is used for enterprise level[14] and commodity linear tape-open (LTO) tape storage media. Because of its high density, barium ferrite has led to data capacity improvements in both enterprise and LTO tapes over prior metal particle (MP) media technology.[15]
Developments in the field have also resulted in the reduction of the size of BaFe particles to about 20 nm. This contrasts with MP technology, which has problems shrinking the particles past 100 nm.[4] Barium ferrite has better packing properties than most other metal particles because of the distinctive shape of the particles.[4] This leads to better control over magnetic orientation and improved signal-to-noise characteristics.[14]
Natural occurrence
The compound occurs in nature, although is exceedingly rare. It is called barioferrite and is related to pyrometamorphism.[16][17]
References
- ^ ISSN 0079-6425.
- ^ S2CID 98312433.
- S2CID 97566562.
- ^ S2CID 22303270.
- PMID 27185343.
- .
- ISBN 9781681081779.
- ISBN 9781483103174.
- ^ .
- ^ "Characteristics of ferrite magnets". e-Magnets UK. Retrieved 8 December 2013.
- ^ "Barium ferrite: Overview". Fujifilm. Archived from the original on 13 August 2017. Retrieved 13 August 2017.
- ISBN 9780750644730.
- ^ "Hard ferrite (ceramic) magnets". Magnaworks Technology. Archived from the original on 20 October 2018. Retrieved 8 December 2013.
- ^ a b "Barium ferrite: Overview". Data storage. Fujifilm Technologies. United States: Fujifilm.
- ^ "FujiFilm barium-ferrite magnetic tape establishes world record in data density: 29.5 billion bits per square inch" (Press release). Fujifilm. 22 January 2010. Archived from the original on 19 October 2020. Retrieved 12 October 2020.
- ^ "Barioferrite". mindat.org.
- ^ "List of Minerals". ima-mineralogy.org. 21 March 2011.

![{\displaystyle {\ce {->[{\text{Δ}}]}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/75d5d7f52b701b6c4f9d85e22372bcec275983ee)