Barium ferrate

| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Barium ferrate(VI)
| |||
| Other names
Barium ferrate(2-)
| |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
| |||
| |||
| Properties | |||
| BaFeO4 | |||
| Molar mass | 257,1646 g/mol | ||
| Appearance | Dark red, opaque crystals | ||
| insoluble | |||
| Structure | |||
| orthorhombic | |||
| Pnma, No. 62[1] | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Barium ferrate is the chemical compound of formula BaFeO4. This is a rare compound containing iron in the +6 oxidation state.[2] The ferrate(VI) ion has two unpaired electrons, making it paramagnetic.[3] It is isostructural with BaSO4, and contains the tetrahedral [FeO4]2− anion.[4]
Structure
The ferrate(VI) anion is paramagnetic due to its two unpaired electrons and it has a tetrahedral molecular geometry.[3]
X-ray diffraction has been used to determine the orthorhombic unit cell structure[1] (lattice vectors a ≠ b ≠ c, interaxial angles α=β=γ=90°)[5] of nanocrystalline BaFeO4. It crystallized in the Pnma space group (point group: D2h) with lattice parameters a = 0.8880 nm, b = 0.5512 nm and c = 0.7214 nm.[1] The accuracy of the X-Ray diffraction data has been verified by the lattice fringe intervals from High-Resolution Transmission Electron Microscopy (HRTEM) and cell parameters calculated from Selected Area Diffraction (SAED).[1]

Characterization
Infrared absorbance peaks of barium ferrate are observed at 870, 812, 780 cm−1.[7]
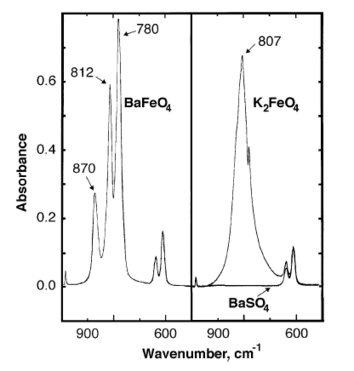
BaFeO4 follows the
Preparation and chemistry
Barium ferrate(VI) can be prepared by both wet and dry synthetic methods. Dry synthesis is usually performed using a thermal technique,[7] such as by heating barium hydroxide and iron(II) hydroxide in the presence of oxygen to about 800 to 900 °C.[10]
- Ba(OH)
2 + Fe(OH)
2 + O
2 → BaFeO
4 + 2 H
2O
Wet methods employ both chemical and electrochemical techniques. For example, the ferrate anion forms when a suitable iron
- 2 Fe(OH)
3 + 3 OCl−
+ 4 OH−
→ 2 FeO2−
4 + 5 H
2O + 3 Cl−
Barium ferrate is then precipitated from solution by adding a solution of a
Uses
Barium ferrate is an oxidizing agent and is used as an oxidizing reagent in organic syntheses. Its other applications include removal of color, removal of cyanide, killing bacteria and contaminated and waste water treatment.[7]
Salts of ferrate(VI) are energetic cathode materials in "super-iron" batteries. Cathodes containing ferrate(VI) compounds are referred to as "super-iron" cathodes due to their highly oxidized iron basis, multiple electron transfer, and high intrinsic energy. Among all ferrate(VI) salts, barium ferrate sustains unusually facile charge transfer, which is important for the high power domain of alkaline batteries.[8]
Reactions
Barium ferrate is the most stable of the ferrate(VI) compounds. It can be prepared in its purest state and has the most definite composition. Barium ferrate can be easily decomposed by all soluble acids, including carbonic acid. If carbon dioxide is passed through water on which hydrated barium ferrate is suspended, barium ferrate will decompose completely to form barium carbonate, ferric hydroxide and oxygen gas. Alkaline sulfates decompose barium ferrate that has not been dried, forming barium sulfate, ferric hydroxide and oxygen gas.
See also
References
- ^ .
- ISBN 978-981-4105-08-8.
- ^ ISBN 978-0-12-352651-9.
- ISBN 978-0-19-855370-0.
- ^ "IUCr". www.iucr.org. Retrieved 2016-04-29.
- ISBN 9780444595539.
- ^ ISSN 0250-5460.
- ^ .
- .
- ISBN 9788183562232.
- ^ ISBN 9780935702668.
- ^ ISSN 0003-2700.
- .


