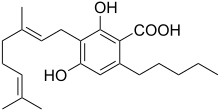
Cannabigerolic acid
| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
3-[(2E)-3,7-Dimethylocta-2,6-dien-1-yl]-2,4-dihydroxy-6-pentylbenzoic acid | |
| Identifiers | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
| Properties | |
| C22H32O4 | |
| Molar mass | 360.494 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Cannabigerolic acid (CBGA) is the acidic form of
phytocannabinoid. It derives from an olivetolic acid. It is a conjugate acid of a cannabigerolate.[1]
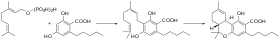
In the Cannabis plant, olivetolic acid and
geranyl diphosphate are synthesized into CBGA.[2]: 6 [3]: 17 CBGA is converted in the plant by CBCA synthase, CBDA synthase and THCA synthase into CBCA, CBDA and THCA respectively.[2]: 6–7 Afterwards, THCA and CBDA can be decarboxylated into THC and CBD by drying and heating plant material. CBGA has emerging pharmacological properties; for example, it had anticonvulsant effects in a mouse model of Dravet syndrome, a form of epilepsy.[4]
COVID-19
In an analysis by the
phytocannabinoids it was found that CBGA had the 2nd highest 3C-like protease inhibitor activity against COVID-19 out of all the phytocannabinoids tested within that study but not as high as the antiviral drug GC376 (72% CBGA vs 24% CBG vs 100% GC376). [5]
A 2022 pre-clinical study by researchers from the
orthosteric binding.[6]
References
- ^ "Cannabigerolic acid". PubChem. United States National Library of Medicine. Retrieved April 7, 2020.
- ^ ISBN 978-0-12-804670-8.
- ISBN 978-0128008270.
- S2CID 236997090.
- PMID 36144858.
- PMID 35007072.
Further reading
- Jacqueline H (9 September 2019). "What is CBGA (Cannabigerolic Acid) & what does this cannabinoid do?". Leafly.
