Carboximidate
This article needs additional citations for verification. (August 2022) |

Carboximidates (or more general imidates) are organic compounds, which can be thought of as esters formed between a imidic acid (R-C(=NR')OH) and an alcohol, with the general formula R-C(=NR')OR".
They are also known as imino ethers, since they resemble imines (>C=N-) with an oxygen atom connected to the carbon atom of the C=N double bond.[1]
Synthesis
Imidates may be generated by a number of synthetic routes,[2] but are in general formed by the Pinner reaction. This proceeds via the acid catalyzed attack of nitriles by alcohols.

Imidates produced in this manner are formed as their hydrochloride salts, which are sometimes referred to as Pinner salts. Carboximidates are also formed as intermediates in the Mumm rearrangement and the Overman rearrangement.
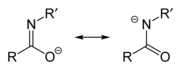
Imidate/amidate anions
An amidate/imidate anion is formed upon
Reactions
Carboximidates are good

Chapman rearrangement
The Chapman rearrangement is the thermal conversion of aryl N‐arylbenzimidates to the corresponding amides, via intramolecular migration of an aryl group from oxygen to nitrogen.[4] It is named after Arthur William Chapman, who first described it,[5] and is conceptually similar to the Newman–Kwart rearrangement.

As a protecting group
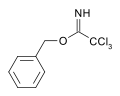
Carboximidates can act as
See also
References
- ^ "Pinner Reaction". Organic Chemistry Portal. Buckten, CH: Reto Mueller. Retrieved 2023-09-26.
- ^ .
- ISBN 978-0-471-22854-7, S. 516.
- ISBN 0471264180.
- .
- ISBN 978-0-471-69754-1.
- .
