Criegee oxidation
| Criegee oxidation | |
|---|---|
| Named after | Rudolf Criegee |
| Reaction type | Organic redox reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000257 |
The Criegee oxidation is a

The rate of the reaction is highly dependent on the relative geometric position of the two
Mechanism
Two mechanisms are proposed for the Criegee oxidation, depending on the configuration of the diol.[6][7] If the oxygen atoms of the two hydroxy groups are conformationally close enough to form a five-membered ring with the lead atom, the reaction occurs via a cyclic intermediate. If the structure cannot adopt such a conformation, an alternate mechanism is possible, but is slower.[8] Trans-fused five member rings are heavily strained, thus trans-diols that are on a five-membered ring will react slower than cis-alcohols on such a structure.[9]
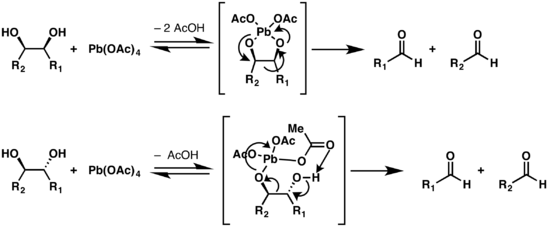
Modifications
Although the classic substrate for the Criegee oxidation are 1,2-diols, the oxidation can be employed with
The Criegee oxidation can also be employed with 2,3-

Criegee oxidations are commonly used in carbohydrate chemistry to cleave 1,2-glycols and differentiate between different kinds of glycol groups.[14]
References
- .
- .
- .
- .
- .
- ISBN 978-0-471-72091-1
- .
- ISBN 0471936235.
- ISBN 978-0-12-429785-2.)
{{cite book}}: CS1 maint: multiple names: authors list (link - .
- .
- .
- .
- PMID 14431883.
