Cyclophane

In
[n]-Cyclophanes
Structures

Paracyclophanes adopt the
Reactivity
With regards to their reactivity, cyclophanes often exhibit diene-like behavior, despite evidence for aromaticity in even the most distorted [6]-cyclophane. This highly distorted cyclophane photochemically converts to the Dewar benzene derivative. Heat reverses the reaction.[5] With
A non-bonding nitrogen to arene distance of 244 pm is recorded for a pyridinophane and in the unusual
NMR properties
The
Synthesis

[6]paracyclophane can be synthesized beginning with the
Metacyclophanes are generally less strained and thus more easily prepared than paracyclophanes. Shown below is the route to a [14][14]metaparacyclophane 4.
![Scheme 4. [14][14]metaparacyclophane](http://upload.wikimedia.org/wikipedia/commons/thumb/9/93/Metaparacyclophane.png/600px-Metaparacyclophane.png)
Naturally occurring [n]-cyclophanes
A few cyclophanes exist in nature. One example of a metacyclophane is cavicularin.
Haouamine A is a paracyclophane found in a certain species of
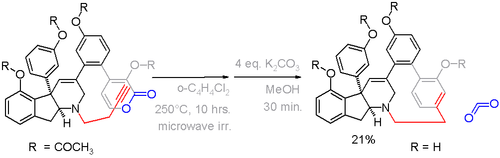
In this compound the deviation from planarity is 13° for the benzene ring and 17° for the bridgehead carbons.[14] An alternative cyclophane formation strategy in scheme 6[15] was developed based on aromatization of the ring well after the formation of the bridge.

Two additional types of cyclophanes were discovered in nature when they were isolated from two species of cyanobacteria from the family Nostocacae.[16] These two classes of cyclophanes are both [7,7] paracyclophanes and were named after the species from which they were extracted: cylindrocyclophanes from Cylindrospermum lichenforme and nostocyclophanes from Nostoc linckia.
[n.n]Paracyclophanes

A well studies member of the [n.n]paracyclophane family is [2.2]paracyclophane.[17][18] One method for its preparation is by the 1,6-Hofmann elimination of 4-methylbenzyltrimethylammonium hydroxide:[19]

The [2.2]paracyclophane-1,9-diene has been applied in

The driving force for ring-opening and polymerization is strain relief. The reaction is believed to be a living polymerization due to the lack of competing reactions.
Because the two benzene rings are in close proximity this cyclophane type also serves as guinea pig for

The product formed has an
Phanes
Generalization of cyclophanes led to the concept of phanes in the IUPAC nomenclature. Some example systematic phane names are:
- [14]metacyclophane is 1(1,3)-benzenacyclopentadecaphane
- [2.2']paracyclophane (or [2.2]paracyclophane) is 1,4(1,4)-dibenzenacyclohexaphane
In "1(1,3)-benzenacyclopentadecaphane", the "1" refers to the first position of the ring as a "superatom", the "(1,3)" describes the "meta" location, "benzena" refers to the ring, and the "pentadeca" (15) describes the chain length counting the ring as one atom.
See also
- Cycloparaphenylene, cyclic all-para-linked phenyl groups.[22]
- Calixarenes
General sources
- B. H. Smith, Bridged Aromatic Compounds, Academic Press, New York, 1964. ISBN 978-0-323-16321-7
- P. M. Keehn, S. M. Rosenfeld (eds.), Cyclophanes, Vols. 1 and 2, Academic Press,New York, 1983. ISBN 978-0-12-403001-5
- F. Vögtle, F., G. Hohner, Top. Curr. Chem. 1978, 74, 1.
- F. Vögtle, P. Neumann, Top. Curr. Chem. 1983, 113, 1; 1985, 115, 1.
References
- ^ ISBN 978-0-471-72091-1
- ISBN 3527603964.
- .
- .
- .
- ^ S2CID 55279751.
- PMID 16089445.
- PMID 20112943.
- PMID 23971948.
- ^ .
- PMID 12662074.
- Ramberg-Bäcklund Reaction with halide donor dibromodifluoromethane and base potassium hydroxide. Final step hydrogenation pf alkene by hydrogen and palladium on carbon
- PMID 16551088. The authors mark the biosynthetic origin as mysterious
- PMID 16623580.
- Nosylate
- .
- PMID 30065985.
- PMID 31283092.
- .
- PMID 17061303.
- PMID 17177393.
- PMID 23126565.
