Exoelectrogen
An exoelectrogen normally refers to a
Utilization of exoelectrogens is currently being researched in the development of
While the exact process in which a cell will reduce an extracellular acceptor will vary from species to species, methods have been shown to involve the use of an
Aside from releasing electrons to an exogenous final electron acceptor, external electron transfer may serve other purposes. First, cells may transfer electrons directly to each other without the need for an intermediary substance. Pelotomaculum thermopropioncum has been observed linked to
In addition to S. oneidensis MR-1, exoelectrogenic activity has been observed in the following strains of bacteria without an exogenous mediator: Shewanella putrefaciens IR-1, Clostridium butyricum, Desulfuromonas acetoxidans, Geobacter metallireducens, Geobacter sulfurreducens, Rhodoferax ferrireducens, Aeromonas hydrophilia (A3), Pseudomonas aeruginosa, Desulfobulbus propionicus, Geopsychrobacter electrodiphilus, Geothrix fermentans, Shewanella oneidensis DSP10, Escherichia coli, Rhodopseudomonas palustris, Brucella anthropi YZ-1, Desulfovibrio desulfuricans, Acidiphilium sp.3.2Sup5, Klebsiella pneumoniae L17, Thermincola sp.strain JR, Pichia anomala.[1]
Extracellular electron transport mechanisms

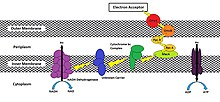
Reduced oxidoreductase enzymes at the extracellular membrane have been shown to use the following methods in transferring their electrons to the exogenous final acceptor: direct contact, shuttling via excreted mediators, iron chelating agents,[9] through a conductive biofilm, and through conductive pili (Figure 2). However, the possibility exists that these methods are not mutually exclusive,[8] and the method used may depend on environmental conditions. Under low microbial population densities, usage of electron shuttles and chelators synthesized by the exoelectrogen may be energetically costly due to insufficient concentrations of such molecules required for recovery and reuse.[9] Under these circumstances, direct transfer would be favored; however, energy benefits would outweigh energy demands when the microbial community is of sufficient size.
Direct reduction of an exogenous acceptor is achieved through contact between the cell’s oxidoreductases and the terminal electron acceptor (i.e. an electrode or external metal compound). While these proteins are diverse (taking on both membrane-bound or soluble forms), their common locations in the outer membrane or periplasm in Gram-negative and Gram-positive bacteria provide intimate contact for electron transfer.[10]
Additionally, the presence of electron shuttles dramatically increases the direct transfer rate.
In the case of Geobacter sulferreducens, the electron carrier riboflavin is used; however, the electron carrier is not entirely freely soluble and can be loosely bound in the culture's biofilm, resulting in a highly conductive biofilm. Furthermore, G. sulferreducens produces electrically conductive pili (nanowires) with OmcS oxidoreductase enzymes embedded on its surface,[12] demonstrating the usage of multiple exoelectrogenic transfer methods.
In iron chelation, insoluble ferric oxide compounds are solubilized in aqueous solutions. As bioavailability of iron is scarce, many microbes secrete iron chelating compounds to solubilize, uptake, and sequester iron for various cellular processes. Certain exoelectrogens have shown capability of using such compounds for electron transport by solubilizing iron extracellularly,[10] and delivering it to the cell surface or within the cell. The components used in each pathway are phylogenetically diverse,[11] thus some chelating agents may reduce iron outside the cell acting as electron shuttles, while others may deliver iron to the cell for membrane bound reduction.[10]
