Ferricyanide
| |
| |
| Names | |
|---|---|
| IUPAC name
iron(3+) hexacyanide
| |
| Systematic IUPAC name
hexacyanidoferrate(III) | |
| Other names
ferric hexacyanide; hexacyanidoferrate(3−); hexacyanoferrate(III)
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
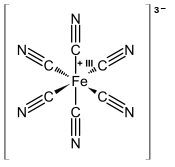
| [Fe(CN)6]3− | |
| Related compounds | |
Other cations
|
Hexacyanonickelate(III)
|
Related compounds
|
Ferrocyanide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ferricyanide is the
oxidant in organic chemistry.[1]
Properties
[Fe(CN)6]3− consists of a Fe3+ center bound in
low spin and easily reduced to the related ferrocyanide
ion [Fe(CN)6]4−, which is a ferrous (Fe2+) derivative. This redox couple is reversible and entails no making or breaking of Fe–C bonds:
- [Fe(CN)6]3− + e− ⇌ [Fe(CN)6]4−
This
redox couple is a standard in electrochemistry
.
Compared to main group cyanides like potassium cyanide, ferricyanides are much less toxic because of the strong bond between the cyanide ion (CN−) and the Fe3+. They do react with mineral acids, however, to release highly toxic hydrogen cyanide gas.
Uses
Treatment of ferricyanide with iron(II) salts affords the brilliant, long-lasting pigment
blueprints
.
See also
References
- ISBN 978-3527306732.
