Liquiritin
Appearance
This article provides insufficient context for those unfamiliar with the subject. (October 2014) |
| |
| Names | |
|---|---|
| IUPAC name
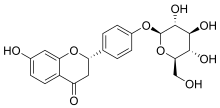
(2S)-4′-(β-D-Glucopyranosyloxy)-7-hydroxyflavan-4-one
| |
| Systematic IUPAC name
(2S)-7-Hydroxy-4-(4-{[(2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)-2,3-dihydro-4H-1-benzopyran-4-one | |
| Other names
Liquiritoside
Liquiritigenin-4'-O-glucoside | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C21H22O9 | |
| Molar mass | 418.398 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Liquiritin is the 4'-O-
licorice.[1]
References
- S2CID 97214921.
Further reading
- Wang J, Wang D, Yu J, Liu C, Li L, Zhang Y (April 2014). PMID 23552847.
- Cong, Jing Xiang; Wang, Shao Yan; Gao, Hong (2012). "Separation of Liquiritin by Two-Dimensional Liquid Chromatography". Advanced Materials Research. 455–456: 1232–1238. S2CID 135570958.
- Cong J, Lin B (March 2007). "Separation of Liquiritin by simulated moving bed chromatography". Journal of Chromatography A. 1145 (1–2): 190–4. PMID 17289063.
- Ni H, Xu M, Xie K, Fei Y, Deng H, He Q, Wang T, Liu S, Zhu J, Xu L, Yao M (2020). "Liquiritin Alleviates Pain Through Inhibiting CXCL1/CXCR2 Signaling Pathway in Bone Cancer Pain Rat". Frontiers in Pharmacology. 11: 436. PMID 32390832.
External links
 Media related to Liquiritin at Wikimedia Commons
Media related to Liquiritin at Wikimedia Commons
