Pinocembrin
| |
| Names | |
|---|---|
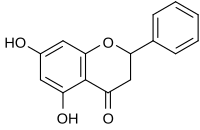
| IUPAC name
5,7-Dihydroxy-2-phenyl-2,3-dihydro-4H-chromen-4-one
| |
| Other names
Dihydrochrysin
Galangin flavanone 5,7-Dihydroxyflavanone 5,7-Dihydroxy-2-phenyl-2,3-dihydro-4H-chromen-4-one 4H-1-Benzopyran-4-one, 2,3-dihydro-5,7-dihydroxy-2-phenyl- 5,7-Dihydroxy-2-phenyl-chroman-4-one | |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H12O4 | |
| Molar mass | 256.257 g·mol−1 |
| Density | 1.386 g/mL |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Pinocembrin is a
Pinocembrin can be converted biosynthetically to pinobanksin by hydroxylation adjacent to the ketone. Studies have shown that pinocembrin has potential as a drug to treat cerebral ischemia, intracerebral hemorrhage, neurodegenerative diseases, cardiovascular diseases and atherosclerosis as well as other diseases.[4][5]
See also
References
External links
 Media related to Pinocembrin at Wikimedia Commons
Media related to Pinocembrin at Wikimedia Commons
