Mercury battery

A mercury battery (also called mercuric oxide battery, mercury cell, button cell, or Ruben-Mallory
For a time during and after World War II, batteries made with
History
The mercury oxide-zinc battery system was known since the 19th century,
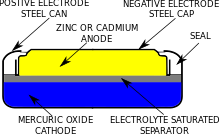
Chemistry
Mercury batteries use either pure mercury(II) oxide (HgO)—also called mercuric oxide—or a mixture of HgO with manganese dioxide (MnO2) as the cathode. Mercuric oxide is a non-conductor, so some graphite is mixed with it; the graphite also helps prevent collection of mercury into large droplets. The half-reaction at the cathode is:[4]
with a
The
followed by the chemical reaction step:[4]
yielding an overall anode half-reaction of:[4]
The overall reaction for the battery is:
In other words, during discharge, zinc is oxidized (loses electrons) to become zinc oxide (ZnO) while the mercuric oxide gets reduced (gains electrons) to form elemental mercury. A little extra mercuric oxide is put into the cell to prevent evolution of hydrogen gas at the end of life.[4]
Electrolyte
Mercuric oxide and cadmium
A different form of mercury battery uses mercuric oxide and cadmium. This has a much lower terminal voltage around 0.9 volts and so has lower energy density, but it has an extended temperature range, in special designs up to 180 C. Because cadmium has low solubility in the alkaline electrolyte, these batteries have long storage life.[4] A 12 volt battery of this type was formerly used for residential smoke detectors. It was designed as a series stack of cells, where one cell had a reduced capacity resulting in a very distinct two-step voltage discharge characteristic. When reaching the end of its life, this smaller cell would discharge first causing the battery terminal voltage to drop sharply by 0.9 volts. This provided a very predictable and repeatable way to warn users the battery needed replacement while the larger capacity cells kept the unit functioning normally.[6]
Electrical characteristics
Mercury batteries using a mercury(II) oxide cathode have a very flat discharge curve, holding constant 1.35 V (open circuit) voltage until about the last 5% of their lifetime, when their voltage drops rapidly. The voltage remains within 1% for several years at light load, and over a wide temperature range, making mercury batteries useful as a voltage reference in electronic instruments and in photographic light meters.[7]
Mercury batteries with cathodes made of a mix of mercuric oxide and manganese dioxide have output voltage of 1.4 V and a more sloped discharge curve.[4]
Product ban
The 1991
In 1992, the state of New Jersey prohibited sales of mercury batteries. In 1996, the United States Congress passed the Mercury-Containing and Rechargeable Battery Management Act that prohibited further sale of mercury-containing batteries (with an exception for up to 25 mg of mercury per button cell). In some specific cases large mercury-containing batteries can continue to be produced if manufacturers provide a system to collect waste batteries and a reclamation facility.[8][9]
Substitutes
The ban on sale of mercury oxide batteries caused numerous problems for
Special adapters with voltage dropping
Use in zinc batteries
Formerly, the zinc anodes of dry cells were amalgamated with mercury, to prevent side-reactions of the zinc with the electrolyte that would reduce the service life of the battery. The mercury took no part in the chemical reaction for the battery. Manufacturers have changed to a purer grade of zinc, so amalgamation is no longer required and mercury is eliminated from the dry cell.
See also
- List of battery types
- List of battery sizes
- Comparison of battery types
- Battery recycling
- Battery nomenclature
References
- ^ ISBN 978-1-4684-9047-3.
- ^ ISBN 0-911937-82-X.
- ^ Clarke, Charles Leigh (1884-06-06). Galvanic battery. US Patent 298175. [1]
- ^ ISBN 0-07-135978-8.
- ^ "Engineering data - Energizer No. E146X" (PDF). Energizer. Archived (PDF) from the original on 2018-11-18. Retrieved 2019-05-11.
- ^ Crompton, Thomas Roy. Battery reference book. pp. 5–23.
- ISBN 0-93557826-9.
- ISBN 0-07-135623-1.
- ^ "IMERC Fact Sheet: Mercury Use in Batteries". Northeast Waste Management Officials' Association. January 2010. Retrieved 2013-06-20.
- ^ Paul, Matthias R. (2009-03-14). "Minolta SR-T Batterieadapter" [Using a 7×7 mm SMD transistor-based low-side voltage regulator circuit as Mercury battery replacement]. Minolta-Forum (in German). Archived from the original on 2016-03-27. Retrieved 2011-02-26.
- ^ a b c Paul, Matthias R. (2005-12-12). "Minolta SR-T Batterieadapter" [Using a Bandgap voltage reference as Mercury battery replacement]. Minolta-Forum (in German). Archived from the original on 2016-10-11. Retrieved 2011-02-26.






