Sodium-ion battery
| Energy density | 250–375 W·h/L |
|---|---|
| Cycle durability | "thousands"[1] of cycles |
| Nominal cell voltage | 3.2-3.6 V |
Sodium-ion batteries (NIBs, SIBs, or Na-ion batteries) are several types of
SIBs received academic and commercial interest in the 2010s and early 2020s, largely due to the uneven geographic distribution, high environmental impact, and high cost of lithium. An obvious advantage of sodium is its natural abundance,
The development of Na+ batteries started in the 1990s. After three decades of development, NIBs are at a critical moment of commercialization. Several companies such as HiNa and CATL in China, Faradion in the United Kingdom, Tiamat in France, Northvolt in Sweden,[5] and Natron Energy in the US, are close to achieving the commercialization of NIBs, with the aim of employing sodium layered transition metal oxides (NaxTMO2), Prussian white (a Prussian blue analogue[6]) or vanadium phosphate as cathode materials.[7]
History
Sodium-ion battery development took place in the 1970s and early 1980s. However, by the 1990s, lithium-ion batteries had demonstrated more commercial promise, causing interest in sodium-ion batteries to decline.[10][11] In the early 2010s, sodium-ion batteries experienced a resurgence, driven largely by the increasing cost of lithium-ion battery raw materials.[10]
Operating principle
SIB cells consist of a cathode based on a sodium-based material, an anode (not necessarily a sodium-based material) and a liquid electrolyte containing dissociated sodium salts in polar protic or aprotic solvents. During charging, sodium ions move from the cathode to the anode while electrons travel through the external circuit. During discharge, the reverse process occurs.
Materials
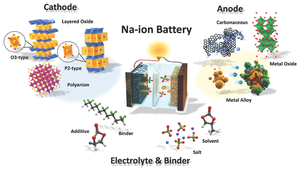
Due to the physical and electrochemical properties of sodium, SIBs require different materials from those used for LIBs.[12]
Anodes
Carbons
SIBs can use
In 2015, researchers demonstrated that graphite could co-intercalate sodium in ether-based electrolytes. Low capacities around 100 mAh/g were obtained with relatively high working potentials between 0 – 1.2 V vs Na/Na+.[17]
One drawback of carbonaceous materials is that, because their intercalation potentials are fairly negative, they are limited to non-aqueous systems.
Plasma-derived Hard Carbon
Plasma-derived hard carbon is an emerging area for SIB batteries.[18] Plasma-derived hard carbon uplift Coulombic efficiency and specific capacity by 33% and 44%. Spark sintering plasma has shown initial Coulombic efficiency of ∼90 % reversible capacity of ∼300 mAh/g and rate capacity of 136.6 mAh/g at 5 A/g. The future aspects of plasma methods to perform multi-material doping, in-situ nanoarchitecture fabrications, and challenges around SIB functioning in extreme environments, and the development of real-time robust monitoring and diagnostic tools to make safe, stable, and high-performance SIB with long life. Further, a data-driven manufacturing framework suggests integrating material informatics with experimental protocols for virtual synthesis of hard carbon; estimating material formulations, manufacturing methods, process-property-performance relationship, and limitations before physical manufacturing of high-performance sodium batteries.[18]
Graphene
Graphene Janus particles have been used in experimental sodium-ion batteries to increase energy density. One side provides interaction sites while the other provides inter-layer separation. Energy density reached 337 mAh/g.[19]
Carbon arsenide
Carbon arsenide (AsC5) mono/bilayer has been explored as an anode material due to high specific gravity (794/596 mAh/g), low expansion (1.2%), and ultra low diffusion barrier (0.16/0.09 eV), indicating rapid charge/discharge cycle capability, during sodium intercalation.[20] After sodium adsorption, a carbon arsenide anode maintains structural stability at 300 K, indicating long cycle life.
Metal alloys
Numerous reports described anode materials storing sodium via alloy reaction and/or conversion reaction.[10] Alloying sodium metal brings the benefits of regulating sodium-ion transport and shielding the accumulation of electric field at the tip of sodium dendrites.[21] Wang, et al. reported that a self-regulating alloy interface of nickel antimony (NiSb) was chemically deposited on Na metal during discharge. This thin layer of NiSb regulates the uniform electrochemical plating of Na metal, lowering overpotential and offering dendrite-free plating/stripping of Na metal over 100 h at a high areal capacity of 10 mAh cm−2.[22]
Metals
Many metals and semi-metals (Pb, P, Sn, Ge, etc.) form stable alloys with sodium at room temperature. Unfortunately, the formation of such alloys is usually accompanied by a large volume change, which in turn results in the pulverization (crumbling) of the material after a few cycles. For example, with tin sodium forms an alloy Na
15Sn
4, which is equivalent to 847 mAh/g specific capacity, with a resulting enormous volume change up to 420%.[23]
In one study, Li et al. prepared sodium and metallic tin Na
15Sn
4/Na through a spontaneous reaction.[24] This anode could operate at a high temperature of 90 °C (194 °F) in a carbonate solvent at 1 mA cm−2 with 1 mA h cm−2 loading, and the full cell exhibited a stable charge-discharge cycling for 100 cycles at a current density of 2C.[24] (2C means that full charge or discharge was achieved in 0.5 hour). Despite sodium alloy's ability to operate at extreme temperatures and regulate dendritic growth, the severe stress-strain experienced on the material in the course of repeated storage cycles limits cycling stability, especially in large-format cells.
Researchers from Tokyo University of Science achieved 478 mAh/g with nano‐sized magnesium particles, announced in December 2020.[25]
Oxides
Some sodium titanate phases such as Na2Ti3O7,[26][27][28] or NaTiO2,[29] delivered capacities around 90–180 mAh/g at low working potentials (< 1 V vs Na/Na+), though cycling stability was limited to a few hundred cycles.
Molybdenum disulphide
In 2021, researchers from China tried layered structure MoS2 as a new type of anode for sodium-ion batteries. A dissolution-recrystallization process densely assembled carbon layer-coated MoS2 nanosheets onto the surface of
Other anodes for Na+
Some other materials, such as
Cathodes
Oxides
Many layered
A P2-type Na2/3Fe1/2Mn1/2O2 oxide from earth-abundant Fe and Mn resources can reversibly store 190 mAh/g at average discharge voltage of 2.75 V vs Na/Na+ utilising the Fe3+/4+ redox couple – on par or better than commercial lithium-ion cathodes such as LiFePO4 or LiMn2O4.[33] However, its sodium deficient nature lowered energy density. Significant efforts were expended in developing Na-richer oxides. A mixed P3/P2/O3-type Na0.76Mn0.5Ni0.3Fe0.1Mg0.1O2 was demonstrated to deliver 140 mAh/g at an average discharge voltage of 3.2 V vs Na/Na+ in 2015.[34] In particular, the O3-type NaNi1/4Na1/6Mn2/12Ti4/12Sn1/12O2 oxide can deliver 160 mAh/g at average voltage of 3.22 V vs Na/Na+,[35] while a series of doped Ni-based oxides of the stoichiometry NaaNi(1−x−y−z)MnxMgyTizO2 can deliver 157 mAh/g in a sodium-ion “full cell” with a hard carbon anode at average discharge voltage of 3.2 V utilising the Ni2+/4+ redox couple.[36] Such performance in full cell configuration is better or on par with commercial lithium-ion systems. A Na0.67Mn1−xMgxO2 cathode material exhibited a discharge capacity of 175 mAh/g for Na0.67Mn0.95Mg0.05O2. This cathode contained only abundant elements.[37] Copper-substituted Na0.67Ni0.3−xCuxMn0.7O2 cathode materials showed a high reversible capacity with better capacity retention. In contrast to the copper-free Na0.67Ni0.3−xCuxMn0.7O2 electrode, the as-prepared Cu-substituted cathodes deliver better sodium storage. However, cathodes with Cu are more expensive.[38]
Oxoanions
Research has also considered cathodes based on
Prussian blue and analogues
Numerous research groups investigated the use of Prussian blue and various Prussian blue analogues (PBAs) as cathodes for Na+-ion batteries. The ideal formula for a discharged material is Na2M[Fe(CN)6], and it corresponds to the theoretical capacity of ca. 170 mAh/g, which is equally split between two one-electron voltage plateaus. Such high specific charges are rarely observed only in PBA samples with a low number of structural defects.
For example, the patented rhombohedral Na2MnFe(CN)6 displaying 150–160 mAh/g in capacity and a 3.4 V average discharge voltage[45][46][47] and rhombohedral Prussian white Na1.88(5)Fe[Fe(CN)6]·0.18(9)H2O displaying initial capacity of 158 mAh/g and retaining 90% capacity after 50 cycles.[48]
While Ti, Mn, Fe and Co PBAs show a two-electron electrochemistry, the Ni PBA shows only one-electron (Ni is not electrochemically active in the accessible voltage range). Iron-free PBA Na2MnII[MnII(CN)6] is also known. It has a fairly large reversible capacity of 209 mAh/g at C/5, but its voltage is unfortunately low (1.8 V versus Na+/Na).[49]
Electrolytes
Sodium-ion batteries can use aqueous and non-aqueous electrolytes. The limited electrochemical stability window of water results in lower voltages and limited energy densities. Non-aqueous carbonate ester polar aprotic solvents extend the voltage range. These include ethylene carbonate, dimethyl carbonate, diethyl carbonate, and propylene carbonate. The most widely used salts in non-aqueous electrolytes are NaClO4 and sodium hexafluorophosphate (NaPF6) dissolved in a mixture of these solvents. It is a well-established fact that these carbonate-based electrolytes are flammable, which pose safety concerns in large-scale applications. A type of glyme-based electrolyte, with sodium tetrafluoroborate as the salt is demonstrated to be non-flammable.[50] In addition, NaTFSI (TFSI = bis(trifluoromethane)sulfonimide) and NaFSI (FSI = bis(fluorosulfonyl)imide, NaDFOB (DFOB = difluoro(oxalato)borate) and NaBOB (bis(oxalato)borate) anions have emerged lately as new interesting salts. Of course, electrolyte additives can be used as well to improve the performance metrics.[51]
Comparison
Sodium-ion batteries have several advantages over competing battery technologies. Compared to lithium-ion batteries, sodium-ion batteries have somewhat lower cost, better safety characteristics, and similar power delivery characteristics, but also a lower energy density.
The table below compares how NIBs in general fare against the two established rechargeable battery technologies in the market currently: the lithium-ion battery and the rechargeable
| Sodium-ion battery | Lithium-ion battery | Lead–acid battery | |
|---|---|---|---|
| Cost per kilowatt-hour of capacity | $40–77 (theoretical in 2019)[53] | $137 (average in 2020)[54] | $100–300[55] |
| Volumetric energy density | 250–375 W·h/L, based on prototypes[56] | 200–683 W·h/L[57] | 80–90 W·h/L[58] |
| Gravimetric energy density (specific energy) | 75–200 W·h/kg, based on prototypes and product announcements[56][59][60] | 120–260 W·h/kg (without protective case needed for battery pack in vehicle)[57] | 35–40 Wh/kg[58] |
| Cycles at 80% depth of discharge[a] | Hundreds to thousands[1] | 3,500[55] | 900[55] |
| Safety | Low risk for aqueous batteries, high risk for Na in carbon batteries | High risk[b] | Moderate risk |
| Materials | Earth-abundant | Scarce | Toxic |
| Cycling stability | High (negligible self-discharge) [citation needed] | High (negligible self-discharge) [citation needed] | Moderate (high self-discharge) [citation needed] |
| Direct current round-trip efficiency | up to 92%[1] | 85–95%[61] | 70–90%[62] |
| Temperature range[c] | −20 °C to 60 °C[1] | Acceptable:−20 °C to 60 °C.
Optimal: 15 °C to 35 °C[63] |
−20 °C to 60 °C[64] |
Commercialization
Companies around the world have been working to develop commercially viable sodium-ion batteries. A 2-hour 5MW/10MWh grid battery was installed in China in 2023.[65]
Active
Altris AB
Altris AB was founded by Associate Professor Reza Younesi, his former PhD student, Ronnie Mogensen, and Associate Professor William Brant as a spin-off from Uppsala University, Sweden,[66] launched in 2017 as part of research efforts from the team on sodium-ion batteries. The research was conducted at the Ångström Advanced Battery Centre led by Prof. Kristina Edström at Uppsala University. The company offers a proprietary iron-based Prussian blue analogue for the positive electrode in non-aqueous sodium-ion batteries that use hard carbon as the anode.[67] Altris holds patents on non-flammable fluorine-free electrolytes consisting of NaBOB in alkyl-phosphate solvents, Prussian white cathode, and cell production. Clarios is partnering to produce batteries using Altris technology.[68]
BYD
The BYD Company is a Chinese electric vehicle manufacturer and battery manufacturer. In 2023, they invested $1.4B USD into the construction of a sodium-ion battery plant in Xuzhou with an annual output of 30 GWh.[69]
CATL
Chinese battery manufacturer CATL announced in 2021 that it would bring a sodium-ion based battery to market by 2023.[70] It uses Prussian blue analogue for the positive electrode and porous carbon for the negative electrode. They claimed a specific energy density of 160 Wh/kg in their first generation battery.[59] The company planned to produce a hybrid battery pack that includes both sodium-ion and lithium-ion cells.[71]
Faradion Limited
Faradion Limited is a subsidiary of India's Reliance Industries.[72] Its cell design uses oxide cathodes with hard carbon anode and a liquid electrolyte. Their pouch cells have energy densities comparable to commercial Li-ion batteries (160 Wh/kg at cell-level) with good rate performance till 3C and cycle lives of 300 (100% depth of discharge) to over 1,000 cycles (80% depth of discharge). Its battery packs have demonstrated use for e-bike and e-scooter applications.[36] They demonstrated transporting sodium-ion cells in the shorted state (at 0 V), eliminating risks from commercial transport of such cells.[73] It is partnering with AMTE Power plc[74] (formerly known as AGM Batteries Limited).[75][76][77][78]
In November 2019, Faradion co-authored a report with Bridge India[79] titled ’The Future of Clean Transportation: Sodium-ion Batteries’[80] looking at the growing role India can play in manufacturing sodium-ion batteries.
On December 5, 2022, Faradion installed its first sodium-ion battery for Nation in New South Wales Australia.[81]
HiNA Battery Technology Company
HiNa Battery Technology Co., Ltd is, a spin-off from the Chinese Academy of Sciences (CAS). It leverages research conducted by Prof. Hu Yong-sheng's group at the Institute of Physics at CAS. HiNa's batteries are based on Na-Fe-Mn-Cu based oxide cathodes and anthracite-based carbon anode. In 2023, HiNa partnered with JAC as the first company to put a sodium-ion battery in an electric car, the Sehol E10X. HiNa also revealed three sodium-ion products, the NaCR32140-ME12 cylindrical cell, the NaCP50160118-ME80 square cell and the NaCP73174207-ME240 square cell, with gravimetric energy densities of 140 Wh/kg, 145 Wh/kg and 155 Wh/kg respectively.[82] In 2019, it was reported that HiNa installed a 100 kWh sodium-ion battery power bank in East China.[83]
Chinese automaker Yiwei debuted the first sodium-ion battery-powered car in 2023. It uses JAC Group’s UE module technology, which is similar to CATL's cell-to-pack design.[84] The car has a 23.2 kWh battery pack with a CLTC range of 230 kilometres (140 mi).[85]
KPIT Technologies
KPIT Technologies introduced India’s first sodium-ion battery technology, marking a significant breakthrough in the country. This newly developed technology is predicted to reduce the cost of batteries for electric vehicles by 25-30%. It has been developed in cooperation with Pune's Indian Institute of Science Education and Research over the course of almost a decade and claims several notable benefits over existing alternatives such as lead-acid and lithium-ion. Among its standout features are a longer lifespan of 3,000–6,000 cycles, faster charging than traditional batteries, greater resistance to below-freezing temperatures and with varied energy densities between 100 and 170 Wh/Kg.[86][87][88]
Natron Energy
Natron Energy, a spin-off from Stanford University, uses Prussian blue analogues for both cathode and anode with an aqueous electrolyte.[89] Clarios is partnering to produce a battery using Natron technology.[90]
Northvolt
Northvolt, Europe’s only large homegrown electric battery maker, has said it has made a “breakthrough” sodium-ion battery. Northvolt said its new battery, which has an energy density of more than 160 watt-hours per kilogram, has been designed for electricity storage plants but could in future be used in electric vehicles, such as two wheeled scooters.[5]
TIAMAT
TIAMAT spun off from the
They are responsible for one of the first commercialized product powered by Sodium-Ion battery technology, as of October 2023, through the commercialization of an electric screw-driver.[96]
SgNaPLus
SgNaPlus is a spin off from National University of Singapore, that uses a propeitary electrode and electrolyte. [1] It is based in Singapore and leverages on research conducted by Alternative Energy Systems Laboratory (AESL) from Energy and Bio-Thermal Systems Division in the Department of Mechanical Engineering, National University of Singapore (NUS)[2]. The division is founded by Prof Palani Balaya. SgNaPlus also has rights for the patent for a non-flammable sodium-ion batteries.
Defunct
Aquion Energy
Aquion Energy was (between 2008 and 2017) a spin-off from Carnegie Mellon University. Their batteries (salt water battery) were based on sodium titanium phosphate anode, manganese dioxide cathode, and aqueous sodium perchlorate electrolyte. After receiving government and private loans, the company filed for bankruptcy in 2017. Its assets were sold to a Chinese manufacturer Juline-Titans, who abandoned most of Aquion's patents.[97][98][96]
See also
- List of battery types
- Alkali metal-ion batteries:
- Lithium-ion battery
- Sodium-ion battery
- Potassium-ion battery
- Alkaline earth metal-ion batteries:
- Rechargeable battery
Notes
- ^ The number of charge-discharge cycles a battery supports depends on multiple considerations, including depth of discharge, rate of discharge, rate of charge, and temperature. The values shown here reflect generally favorable conditions.
- ^ See Lithium-ion battery safety.
- ^ Temperature affects charging behavior, capacity, and battery lifetime, and affects each of these differently, at different temperature ranges for each. The values given here are general ranges for battery operation.
References
- ^ a b c d "Performance". Faradion Limited. Retrieved 17 March 2021.
The (round trip) energy efficiency of sodium-ion batteries is 92% at a discharge time of 5 hours.
- .
- ^ Xie M, Wu F, Huang Y. Sodium-ion batteries: Advanced technology and applications: De Gruyter; 2022. 1-376 pp. page 8. doi: 10.1515/9783110749069.
- ^ a b Handbook of Sodium-Ion Batteries. 2023. R.R. Gaddam, G. Zhao. doi: 10.1201/9781003308744.
- ^ a b Lawson, Alex. "'Breakthrough battery' from Sweden may cut dependency on China". The Guardian. Retrieved 22 November 2023.
- S2CID 259615584.
- ^ Sodium-based batteries: development, commercialization journey and new emerging chemistries. 2023. Oxf Op Mater Sci. 3/1. P. Yadav, V. Shelke, A. Patrike, M. Shelke. doi: 10.1093/oxfmat/itac019
* Strategies and practical approaches for stable and high energy density sodium-ion battery: a step closer to commercialization. 2023. Materials Today Sustainability. 22/. P. Yadav, A. Patrike, K. Wasnik, V. Shelke, M. Shelke. doi: 10.1016/j.mtsust.2023.100385
* Chapter 6 The commercialization of sodium-ion batteries. 2022. 306-62. doi: 10.1515/9783110749069-006
* The design, performance and commercialization of Faradion's non-aqueous Na-ion battery technology. 2021. Na-ion Batteries. 313-44. A. Rudola, F. Coowar, R. Heap, J. Barker. doi: 10.1002/9781119818069.ch8
* Non-Aqueous Electrolytes for Sodium-Ion Batteries: Challenges and Prospects Towards Commercialization. 2021. Batteries and Supercaps. 4/6, 881–96. H. Hijazi, P. Desai, S. Mariyappan. doi: 10.1002/batt.202000277
* (Invited) The Scale-up and Commercialization of a High Energy Density Na-Ion Battery Technology. 2019. ECS Meeting Abstracts. MA2019-03/1, 64-. J. Barker. doi: 10.1149/ma2019-03/1/64
* Sodium-Ion Batteries: From Academic Research to Practical Commercialization. 2018. Advanced Energy Materials. 8/4. J. Deng, W.B. Luo, S.L. Chou, H.K. Liu, S.X. Dou. doi: 10.1002/aenm.201701428
* The Scale-up and Commercialization of Nonaqueous Na-Ion Battery Technologies. 2018. Advanced Energy Materials. 8/17, 13. A. Bauer, J. Song, S. Vail, W. Pan, J. Barker, Y. Lu. doi: 10.1002/aenm.201702869 - ^ Hina Battery becomes 1st battery maker to put sodium-ion batteries in EVs in China, CnEVPost, 23 February 2023
- ^ "Pylontech Obtains the World's First Sodium Ion Battery Certificate from TÜV Rheinland". 8 March 2023.
- ^ PMID 28349134.
- PMID 25390643.
- PMID 28627780.
- ISSN 0013-4651.
- ISSN 1099-0062.
- ^ .
- PMID 25056756.
- ^ ISSN 2352-152X.license.
 This article incorporates text from this source, which is available under the CC BY-SA 4.0
This article incorporates text from this source, which is available under the CC BY-SA 4.0 - ^ Lavars, Nick (2021-08-26). "Two-faced graphene offers sodium-ion battery a tenfold boost in capacity". New Atlas. Retrieved 2021-08-26.
- S2CID 260322455.
- ^ "Northwestern SSO". prd-nusso.it.northwestern.edu. Retrieved 2021-11-19.
- S2CID 237307044. Retrieved 2021-11-19.
- ^ Bommier C and Ji X. Recent development on anodes for Na-ionbatteries. Isr J Chem, 2015; 55(5): 486–507.
- ^ a b "Northwestern SSO". prd-nusso.it.northwestern.edu. Retrieved 2021-11-19.
- PMID 33300173.
- ISSN 0897-4756.
- ISSN 2050-7496.
- ISSN 1388-2481.[permanent dead link]
- ISSN 1754-5706.
- S2CID 239640591.
- S2CID 244583592.
- ^ Zhao, Q., Gaddam, R. R., Yang, D., Strounina, E., Whittaker, A. K., and Zhao, X. S. (2018). Pyromellitic dianhydride-based polyimide anodes for sodium-ion batteries. Electrochimica Acta, 265, 702–708.
- PMID 22543301.
- PMID 27134617.
- ^ Kendrick, E.; Gruar, R.; Nishijima, M.; Mizuhata, H.; Otani, T.; Asako, I.; Kamimura, Y. (May 22, 2014). "Tin-Containing Compounds United States Patent No. US 10,263,254" (PDF).
- ^ ISSN 1614-6840.
- .
- .
- hdl:2324/7951.
- ^ Barker, J.; Saidi, Y.; Swoyer, J. L. "Sodium ion Batteries United States Patent No. US 6,872,492 Issued March 29, 2005" (PDF).
- ISSN 1364-5501.
- ISSN 0378-7753.
- ^ Determination of a sodium-ion cell entropy-variation. 2023. Journal of Power Sources. 581/. N. Damay, R. Recoquillé, H. Rabab, J. Kozma, C. Forgez, A. El Mejdoubi, et al. doi: 10.1016/j.jpowsour.2023.233460.
- ^ US20190312299A1, PALANI, Balaya; RUDOLA, Ashish & Du, Kang et al., "Non-flammable sodium-ion batteries", issued 2019-10-10
- S2CID 30623364.
- S2CID 2335024.
- ^ Lu, Y.; Kisdarjono, H.; Lee, J. J.; Evans, D. "Transition metal hexacyanoferrate battery cathode with single plateau charge/discharge curve United States Patent No. 9,099,718 Issued August 4, 2015; Filed by Sharp Laboratories of America, Inc. on October 3, 2013" (PDF).
- S2CID 202881037.
- .
- S2CID 218930265.
- .
- S2CID 206894534.
- .
- ^ "Battery Pack Prices Cited Below $100/kWh for the First Time in 2020, While Market Average Sits at $137/kWh". Bloomberg NEF. 16 December 2020. Retrieved 15 March 2021.
- ^ a b c Mongird K, Fotedar V, Viswanathan V, Koritarov V, Balducci P, Hadjerioua B, Alam J (July 2019). Energy Storage Technology and Cost Characterization Report (PDF) (pdf). U.S. Department Of Energy. p. iix. Retrieved 15 March 2021.
- ^ .
- ^ a b Automotive Li-Ion Batteries: Current Status and Future Perspectives (Report). U.S. Department Of Energy. 2019-01-01. p. 26. Retrieved 15 March 2021.
- ^ ISSN 2352-152X.
- ^ a b "CATL Unveils Its Latest Breakthrough Technology by Releasing Its First Generation of Sodium-ion Batteries". www.catl.com. Retrieved 2023-04-24.
- ^ "CATL to begin mass production of sodium-ion batteries next year". 29 October 2022.
- ^ Lithium Ion Battery Test – Public Report 5 (PDF) (pdf). ITP Renewables. September 2018. p. 13. Retrieved 17 March 2021.
The data shows all technologies delivering between 85–95% DC round-trip efficiency.
- . Retrieved 17 March 2021.
Lead–acid batteries have a ... round trip-efficiency (RTE) of ~70–90%
- S2CID 115675281.
- S2CID 111233540.
- ^ Murray, Cameron (3 August 2023). "'World-first' grid-scale sodium-ion battery project in China enters commercial operation". Energy-Storage.News.
- ^ "Major successes for Uppsala University researchers' battery material – Uppsala University". www.uu.se. 8 June 2022. Retrieved 2023-06-29.
- ^ "Researchers develop electric vehicle battery made from seawater and wood". Electric & Hybrid Vehicle Technology International. 2021-06-17. Retrieved 2021-07-29.
- ^ "Clarios and Altris announce collaboration agreement to advance sustainable sodium-ion battery technology". Default. Retrieved 2024-01-24.
- ^ "BYD & Huaihai move on plans for sodium-ion battery plant". electrive.com. 2023-11-20. Retrieved 2023-11-20.
- ^ "China's CATL unveils sodium-ion battery – a first for a major car battery maker". Reuters. 2021-07-29. Retrieved 2021-11-07.
- ^ Lykiardopoulou, Loanna (2021-11-10). "3 reasons why sodium-ion batteries may dethrone lithium". TNW. Retrieved 2021-11-13.
- ^ "Reliance takes over Faradion for £100 million". electrive.com. 2022-01-18. Retrieved 2022-10-29.
- ^ WO2016027082A1, Barker, Jeremy & Wright, Christopher John, "Storage and/or transportation of sodium-ion cells", issued 2016-02-25 Filed by Faradion Limited on August 22, 2014.
- ^ "Faradion announces a collaboration and licensing deal with AMTE Power". Faradion. 2021-03-10. Retrieved 2021-11-07.
- ^ "Ultra Safe AMTE A5" (PDF). May 2020. Archived from the original (PDF) on 2020-09-27. Retrieved 2021-10-14.
- ^ "Dundee in running as battery cell pioneer AMTE Power closes in on UK 'gigafactory' site". www.scotsman.com. 5 October 2021. Retrieved 2021-11-07.
- S2CID 233516956.
- ^ The Tesla Domain (November 6, 2022), This UK based sodium battery threatens to change the EV industry forever!!, retrieved 2022-11-27
- ^ India, Bridge. "Bridge India Homepage". bridgeindia.org.uk. Bridge India. Retrieved 17 August 2023.
- ^ Rudola, Ashish (24 November 2019). "The Future of Clean Transportation: Sodium-ion Batteries". bridgeindia.org.uk. Bridge India, Faradion. Retrieved 17 August 2023.
- ^ "First Faradion battery installed in Australia". 5 December 2022.
- ^ "Hina Battery Becomes 1st Battery Maker to Put Sodium-ion Batteries in Evs in China". batteriesnews.com. 23 February 2023. Retrieved 2023-02-23.
- ^ "Sodium-ion Battery Power Bank Operational in East China—Chinese Academy of Sciences". english.cas.cn. Retrieved 2019-09-05.
- ^ Johnson, Peter (2023-12-27). "Volkswagen-backed EV maker rolls out first sodium-ion battery powered electric car". Electrek. Retrieved 2023-12-31.
- ^ McDee, Max (6 January 2024). "JAC Group delivers first EVs with sodium-ion battery". ArenaEV. Retrieved 11 January 2024.
- ^ "KPIT Tech launches sodium-ion battery tech". December 13, 2023 – via The Economic Times - The Times of India.
- ^ "KPIT rolls out India's first sodium-ion battery tech, aims at revenue within a year". Moneycontrol. December 13, 2023.
- ^ "KPIT Tech shares zoom; here's what's powering the upmove". Zee Business. December 13, 2023.
- ^ Patel, Prachi (2021-05-10). "Sodium-Ion Batteries Poised to Pick Off Large-Scale Lithium-Ion Applications". IEEE Spectrum. Retrieved 2021-07-29.
- ^ "Natron Collaborates With Clarios on Mass Manufacturing of Sodium-Ion Batteries". Default. Retrieved 2024-01-24.
- ^ "Sodium to boost batteries by 2020". 2017 une année avec le CNRS. 2018-03-26. Archived from the original on 2020-04-18. Retrieved 2019-09-05.
- S2CID 106396927.
- ISSN 1754-5692.
- ^ Hall, N.; Boulineau, S.; Croguennec, L.; Launois, S.; Masquelier, C.; Simonin, L. (October 13, 2015). "Method for preparing a Na3V2(PO4)2F3 particulate material United States Patent Application No. 2018/0297847" (PDF).
- ^ "Tiamat |".
- ^ a b "Public announcement for commercializaton of sodium-ion batteries". Retrieved 2023-11-29.
- ^ "Aqueous electrolyte energy storage device".
- ^ "Large format electrochemical energy storage device housing and module".
External links
- Ma, Bingyuan; Lee, Youngju; Bai, Peng (2021). "Dynamic Interfacial Stability Confirmed by Microscopic Optical Operando Experiments Enables High-Retention-Rate Anode-Free Na Metal Full Cells". Advanced Science. 8 (12): 2005006. PMID 34194939.
- Wunderlich-Pfeiffer, Frank (April 19, 2023). "Na-ion: A battery worth its salt?". intercalationstation.substack.com. Retrieved 2023-04-28.
- Wu, Billy (January 3, 2024). Sodium ion batteries - The low-cost future of energy storage? (Podcast). Retrieved 2024-01-05.

