Nafion

| |
| Identifiers | |
|---|---|
| ChemSpider |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| C7HF13O5S . C2F4 | |
| Molar mass | See Article |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H319, H335 | |
| P261, P264, P271, P280, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233, P405, P501 | |
| Related compounds | |
Related compounds
|
Aciplex Flemion Dowew fumapem F |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Nafion is a brand name for a sulfonated
The chemical basis of Nafion's ion-conductive properties remain a focus of extensive research.
Nomenclature and molecular weight
Nafion can be produced as both a powder
- From Chemical Abstracts: ethanesulfonyl fluoride, 2-[1-[difluoro-[(trifluoroethenyl)oxy]methyl]-1,2,2,2-tetrafluoroethoxy]-1,1,2,2,-tetrafluoro-, with tetrafluoroethylene
- tetrafluoroethylene-perfluoro-3,6-dioxa-4-methyl-7-octenesulfonic acid copolymer
The
Preparation
Nafion derivatives are first synthesized by the copolymerization of tetrafluoroethylene (TFE) (the monomer in Teflon) and a derivative of a perfluoro (alkyl vinyl ether) with sulfonyl acid fluoride. The latter reagent can be prepared by the pyrolysis of its respective oxide or carboxylic acid to give the olefinated structure.[6]
The resulting product is an -SO2F-containing thermoplastic that is extruded into films. Hot aqueous NaOH converts these sulfonyl fluoride (-SO2F) groups into sulfonate groups (-SO3−Na+). This form of Nafion, referred to as the neutral or salt form, is finally converted to the acid form containing the sulfonic acid (-SO3H) groups. Nafion can be dispersed into solution by heating in aqueous alcohol at 250 °C in an autoclave for subsequent casting into thin films or use as polymeric binder in electrodes. By this process, Nafion can be used to generate composite films, coat electrodes, or repair damaged membranes.[3]
Properties
The combination of the stable PTFE backbone with the acidic sulfonic groups gives Nafion its characteristics:[2][7]
- It is highly conductive to cations, making it suitable for many membrane applications.[2]
- It resists chemical attack. According to Chemours, only alkali metals (particularly sodium) can degrade Nafion under normal temperatures and pressures.
- The PTFE backbone interlaced with the ionic sulfonate groups gives Nafion a high chemical stability temperature (e.g. 190 °C) but a softening point in the range of 85-100 °C give it a moderate operating temperature, e.g. up to 100 °C, with additional challenges in all applications due to the loss of water above 100 °C.
- It is a trifluoromethanesulfonic acid, CF3SO3H, although Nafion is a weaker acid by at least three orders of magnitude.
- It is selectively and highly permeable to water.
- Its proton conductivity up to 0.2 S/cm depending on temperature, hydration state, thermal history and processing conditions.[9][2]
- The solid phase and the aqueous phase of Nafion are both permeable to gases,[10][11] which is a drawback for energy conversion devices such as artificial leaves, fuel cells, and water electrolyzers.
Structure/morphology
The morphology of Nafion membranes is a matter of continuing study to allow for greater control of its properties. Other properties such as water management, hydration stability at high temperatures, electro-osmotic drag, as well as the mechanical, thermal, and oxidative stability, are affected by the Nafion structure. A number of models have been proposed for the morphology of Nafion to explain its unique transport properties.[2]

The first model for Nafion, called the cluster-channel or cluster-network model, consisted of an equal distribution of sulfonate ion clusters (also described as 'inverted
The difficulty in determining the exact structure of Nafion stems from inconsistent solubility and crystalline structure among its various derivatives. Advanced morphological models have included a core-shell model where the ion-rich core is surrounded by an ion poor shell, a rod model where the sulfonic groups arrange into crystal-like rods, and a sandwich model where the polymer forms two layers whose sulfonic groups attract across an aqueous layer where transport occurs.[4] Consistency between the models include a network of ionic clusters; the models differ in the cluster geometry and distribution. Although no model has yet been determined fully correct, some scientists have demonstrated that as the membrane hydrates, Nafion's morphology transforms from the cluster-channel model to a rod-like model.[4]
A cylindrical-water channel model[13] was also proposed based on simulations of small-angle X-ray scattering data and solid state nuclear magnetic resonance studies. In this model, the sulfonic acid functional groups self-organize into arrays of hydrophilic water channels, each ~ 2.5 nm in diameter, through which small ions can be easily transported. Interspersed between the hydrophilic channels are hydrophobic polymer backbones that provide the observed mechanical stability. Many recent studies, however, favored a phase-separated nanostructure consisting of locally-flat, or ribbon-like, hydrophilic domains based on evidence from direct-imaging studies[14] and more comprehensive analysis of the structure and transport properties.[2][15]
Applications
Nafion's properties make it suitable for a broad range of applications. Nafion has found use in fuel cells, electrochemical devices, chlor-alkali production, metal-ion recovery, water electrolysis, plating, surface treatment of metals, batteries, sensors, Donnan dialysis cells, drug release, gas drying or humidification, and superacid catalysis for the production of fine chemicals.[3][4][7][16] Nafion is also often cited for theoretical potential (i.e., thus far untested) in a number of fields. With consideration of Nafion's wide functionality, only the most significant will be discussed below.
Chlor-alkali production cell membrane
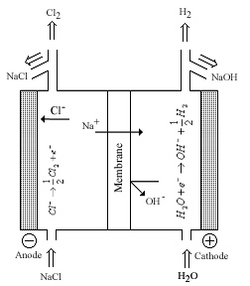
Chlorine and sodium/potassium hydroxide are among the most produced commodity chemicals in the world. Modern production methods produce Cl2 and NaOH/KOH from the electrolysis of brine using a Nafion membrane between half-cells. Before the use of Nafion, industries used mercury containing sodium amalgam to separate sodium metal from cells or asbestos diaphragms to allow for transfer of sodium ions between half cells; both technologies were developed in the latter half of the 19th century. The disadvantages of these systems is worker safety and environmental concerns associated with mercury and asbestos, economical factors also played a part, and in the diaphragm process chloride contamination of the hydroxide product. Nafion was the direct result of the chlor-alkali industry addressing these concerns; Nafion could tolerate the high temperatures, high electrical currents, and corrosive environment of the electrolytic cells.[3][4][7]
The figure to the right shows a chlor-alkali cell where Nafion functions as a membrane between half cells. The membrane allows sodium ions to transfer from one cell to the other with minimal electrical resistance. The membrane was also reinforced with additional membranes to prevent gas product mixing and minimize back transfer of Cl− and −OH ions.[3]
Proton exchange membrane (PEM) for fuel cells
Although fuel cells have been used since the 1960s as power supplies for satellites, recently they have received renewed attention for their potential to efficiently produce clean energy from hydrogen. Nafion was found effective as a membrane for
Superacid catalyst for fine chemical production
Nafion, as a
Alkylation with alkyl halides
Nafion-H gives efficient conversion whereas the alternative method, which employs
Acylation
The amount of Nafion-H needed to catalyze the acylation of benzene with aroyl chloride is 10–30% less than the Friedel-Crafts catalyst:[17]
Catalysis of protection groups
Nafion-H increases
Isomerization
Nafion can catalyze a 1,2-hydride shift.[16]
It is possible to immobilize
Sensors
Nafion has found use in the production of sensors, with application in ion-selective, metallized, optical, and biosensors. What makes Nafion especially interesting is its demonstration in biocompatibility. Nafion has been shown to be stable in cell cultures as well as the human body, and there is considerable research towards the production of higher sensitivity glucose sensors.[3]
Antimicrobial surfaces
Nafion surfaces show an exclusion zone against bacteria colonization.[18] Moreover, layer-by-layer coatings comprising Nafion show excellent antimicrobial properties.[19]
Dehumidification in spacecraft
The SpaceX Dragon 2 human-rated spacecraft uses Nafion membranes to dehumidify the cabin air. One side of the membrane is exposed to the cabin atmosphere, the other to the vacuum of space. This results in dehumidification since Nafion is permeable to water molecules but not air. This saves power and complexity since cooling is not required (as needed with a condensing dehumidifier), and the removed water is rejected to space with no additional mechanism needed.[20]
Modified Nafion for PEM fuel cells
Normal Nafion will dehydrate (thus lose proton conductivity) when the temperature is above ~80 °C. This limitation troubles the design of fuel cells because higher temperatures are desirable for better efficiency and CO tolerance of the platinum catalyst. Silica and zirconium phosphate can be incorporated into Nafion water channels through in situ chemical reactions to increase the working temperature to above 100 °C.
References
- The News Journal. p. B7.
- ^ PMID 28112903.
- ^ .
- ^ PMID 15669162.
- ^ a b "nafion membrane, chemours nafion, proton exchange membrane". www.nafion.com. Retrieved 2021-04-22.
- ^ Connolly, D.J.; Longwood; Gresham, W. F. (1966). "Fluorocarbon Vinyl Ether Polymers". U.S. patent 3,282,875.
{{cite journal}}: Cite journal requires|journal=(help) - ^ a b c Perma Pure LLC (2004). "Nafion: Physical and Chemical Properties". Technical Notes and Articles. Archived from the original on September 28, 2013.
- ISSN 1155-4339. Archived from the original on 2007-06-11.)
{{cite journal}}: CS1 maint: bot: original URL status unknown (link) CS1 maint: multiple names: authors list (link - ISSN 1945-7111.
- .
- .
- .
- PMID 18066069.
- PMID 35596390.
- S2CID 94579140.
- ^ .
- ^ ISBN 978-0-470-01754-8.
- PMID 29132042.
- PMID 31689966.
- ^ Jason Silverman; Andrew Irby; Theodore Agerton (2020). Development of the Crew Dragon ECLSS (PDF). International Conference on Environmental Systems.
External links
- What Nafion Membrane is Right for an Electrolyzer / Hydrogen Generation?
- Homepage of Walther G. Grot
- Walther G. Grot: "Fluorinated Ionomers"
- Isotopic effects on Nafion conductivity
- Membrane thickness on conductivity_of_Nafion
- Nafion hydration
- Nafion Totally Explained at the Wayback Machine (archived 22 September 2007)




