Norrish reaction
A Norrish reaction, named after
Type I
The Norrish type I reaction is the photochemical cleavage or
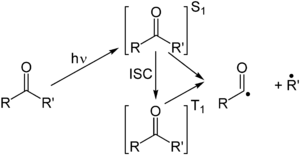
Several secondary reaction modes are open to these fragments depending on the exact molecular structure.
- The fragments can simply recombine to the original carbonyl compound, with racemisationat the α-carbon.
- The acyl radical can lose a molecule of α substituents. Typically the more α substituted a ketone is, the more likely the reaction will yield products in this way.[5][6]
- The abstraction of an α-proton from the carbonyl fragment may form a ketene and an alkane.
- The abstraction of a β-proton from the alkyl fragment may form an aldehyde and an alkene.

The synthetic utility of this reaction type is limited, for instance it often is a side reaction in the Paternò–Büchi reaction. One organic synthesis based on this reaction is that of bicyclohexylidene.[7]
Type II
A Norrish type II reaction is the photochemical

Secondary reactions that occur are fragmentation (β-scission) to form an alkene and an enol (which will rapidly tautomerise to a carbonyl), or intramolecular recombination of the two radicals to a substituted cyclobutane (the Norrish–Yang reaction).[10]
Scope
The Norrish reaction has been studied in relation to
In one study radical.

Leo Paquette's 1982 synthesis of dodecahedrane involves three separate Norrish-type reactions in its approximately 29-step sequence.
An example of a synthetically useful Norrish type II reaction can be found early in the total synthesis of the biologically active

See also
- Photo-Fries rearrangement- a related reaction of aromatic carbonyls
- McLafferty rearrangement - similar to a Type II Norrish reaction. Caused by electron impact ionization rather than light
- Carbon monoxide-releasing molecules
References
- ISBN 0-470-01041-X
- S2CID 225243217.
- ^ doi:10.1351/goldbook.N04219. Retrieved 31 March 2014.)
{{cite journal}}: Cite journal requires|journal=(help - .
- .
- .
- ^ Bicyclohexylidene Nicholas J. Turro, Peter A. Leermakers, and George F. Vesley Organic Syntheses, Coll. Vol. 5, p.297 (1973); Vol. 47, p.34 (1967) Online article.
- doi:10.1351/goldbook.N04218. Retrieved 31 March 2014.)
{{cite journal}}: Cite journal requires|journal=(help - S2CID 4104669.
- doi:10.1351/goldbook.NT07427. Retrieved 31 March 2014.)
{{cite journal}}: Cite journal requires|journal=(help - ^ Technically Au3+ is reduced to Au2+ which then forms Au+ and Au3+ by disproportionation followed by final reduction of Au1+ to Auo
- PMID 23288535.
