Carbon monoxide-releasing molecules
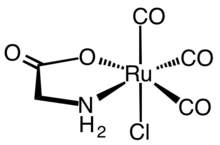
Carbon monoxide-releasing molecules (CORMs) are chemical compounds designed to release controlled amounts of carbon monoxide (CO). CORMs are being developed as potential therapeutic agents to locally deliver CO to cells and tissues, thus overcoming limitations of CO gas inhalation protocols.
CO is best known for its toxicity in carbon monoxide poisoning at high doses. However, CO is a gasotransmitter and supplemental low dosage of CO has been linked to therapeutic benefits. Pre-clinical research has focused on CO's anti-inflammatory activity with significant applications in cardiovascular disease, oncology, transplant surgery, and neuroprotection.[1]
History
Therapeutic interest in CO dates back to the study of
Nickel tetracarbonyl was the first carbonyl-complex used to achieve local delivery of CO and was the first CO delivery molecule suggested to have therapeutic potential in 1891.[2] The acronym CORM was coined in 2002, which marks the first modern biomedical and pharmaceutical initiative.[3] The enzymatic reaction of heme oxygenase inspired the development of synthetic CORMs.
The first synthetic CORMs were typically
CORM classifications
Transition metal CORMs
The majority of therapeutically relevant CORMs are transition metal complexes primarily based on iron, molybdenum, ruthenium, manganese, cobalt, rhenium and others.[6]
PhotoCORMs
The release of CO from carrier agents can be induced photochemically. These carriers are called photoCORMs and include both metal complexes and metal-free (organic) compounds of various structural motifs which could be regarded as a special type of photolabile protecting group.[7]
ET-CORMs
Enzyme-triggered CORMs (ET-CORMs) have been developed to improve selective local delivery of CO. Some ET-CORM prodrugs are activated by esterase enzymes for site specific liberation of CO.[8]
CO prodrugs
Organic CORMs are being developed to overcome reactivity and certain toxicity limitations of inorganic CORMs.
In 2003, cyclic oxocarbons were suggested as a source for therapeutic CO including deltic acid, squaric acid, croconic acid, and rhodizonic acid and their salts.[9]
Recent years have seen increasing interests in organic CO prodrugs because of the need to consider drug developability issues in developing CO-based therapeutics.[10] These CO prodrugs have tunable release rate, triggered release, and the ability to release more than one payload from a single prodrug.[11]
Enzyme hybrids
Based on the synergism of the
Carbon monoxide releasing materials
Carbon monoxide releasing materials (CORMAs) are essentially novel drug formulations and drug delivery platforms which have emerged to overcome the pharmaceutical limitations of most CORM species.
Other advanced drug delivery devices, such as encapsulated CORMs and extracorporeal membrane-inspired technologies, have been developed.[5]
Carboxyhemoglobin infusion
Porphyrins
Porphyrin structures such as heme, hemin, and metallic protoporphyrin IX (PPIX) analogs (such as cobalt PPIX) have been deployed to induce heme oxygenase and subsequently undergo biotransformation to liberate CO, the inorganic ion, and biliverdin/bilirubin.[17] Some PPIX analogs such as tin PPIX, tin mesoporphyrin, and zinc PPIX, are heme oxygenase inhibitors.
Endogenous CO
HMOX is regarded as the main source of endogenous CO production, though other minor contributors have been identified in recent years.[18] CO is formed at a rate of 16.4 μmol/hour in the human body, ~86% originating from heme via heme oxygenase and ~14% from non-heme sources including: photooxidation, lipid peroxidation, and xenobiotics.[19] The average carboxyhemoglobin (CO-Hb) level in a non-smoker is under 3% CO-Hb (whereas a smoker may reach levels near 10% CO-Hb),[20] though geographic location, occupation, health and behavior are contributing variables.
Heme oxygenase
In the late 1960s Rudi Schmid characterized the enzyme that facilitates the reaction for heme catabolism, thereby identifying the heme oxygenase (HMOX) enzyme.
HMOX catalyzes the degradation of heme to
The enzymatic velocity and catalytic activity of HMOX can be enhanced by a plethora of dietary substances and xenobiotics to increase CO production.
Minor CO sources
The formation of CO from
CO pharmacology
Signaling
The first evidence of CO as a signaling molecule occurred upon observation of CO stimulating
CO may have an inhibitory effect on numerous proteins including cytochrome P450 and cytochrome c oxidase.[28]
Pharmacokinetics
CO has approximately 210x greater affinity for hemoglobin than oxygen. The equilibrium dissociation constant for the reaction Hb-CO ⇌ Hb + CO strongly favours the CO complex, thus the release of CO for pulmonary excretion generally takes some time.
Based on this binding affinity, blood is essentially an irreversible sink for CO and presents a therapeutic challenge for the delivery of O2 to cells and tissues.
CO is considered non-reactive in the body and primarily undergoes
References
- S2CID 205477130.
- ^ S2CID 233205099.
- S2CID 12515186.
- PMID 30007887.
- ^ PMID 34481027.
- PMID 24628281.
- PMID 33125209.
- PMID 25009775.
- PMID 17443255.
- PMID 26869408.
- PMID 29762011.
- S2CID 221219782.
- PMID 24556640.
- PMID 33429521.
- PMID 27808304.
- S2CID 51712930.
- S2CID 22652094.
- ^ S2CID 224824871.
- ISBN 978-1-4200-4101-9.
- ISBN 978-1-4160-3406-3.
- ISBN 978-3-319-06150-4.
- PMID 18289074.
- ISBN 978-0-8493-1041-6.
- PMID 15625852.
- ^ Nishibayashi H, Omma T, Sato R, Estabrook RW, Okunuki K, Kamen MD, Sekuzu I, eds. (1968). Structure and Function of Cytochromes. review article. University Park Press. pp. 658–665.
- S2CID 21861993.
- PMID 21521759.
- ISBN 978-0-306-48324-0.
- PMID 23946966.
Further reading
- Kim HH, Choi S (August 2018). "Therapeutic Aspects of Carbon Monoxide in Cardiovascular Disease". review article. International Journal of Molecular Sciences. 19 (8): 2381. PMID 30104479.
- Ismailova A, Kuter D, Bohle DS, Butler IS (2018). "An Overview of the Potential Therapeutic Applications of CO-Releasing Molecules". review article. Bioinorganic Chemistry and Applications. 2018: 8547364. PMID 30158958.
- Abeyrathna N, Washington K, Bashur C, Liao Y (October 2017). "Nonmetallic carbon monoxide releasing molecules (CORMs)". review article. Organic & Biomolecular Chemistry. 15 (41): 8692–8699. PMID 28948260.
- Hopper CP, Wollborn J (August 2019). "Delivery of carbon monoxide via halogenated ether anesthetics". review article. Nitric Oxide. 89: 93–95. S2CID 164217698.
- Wilson JL, Jesse HE, Poole RK, Davidge KS (May 2012). "Antibacterial effects of carbon monoxide". review article. Current Pharmaceutical Biotechnology. 13 (6): 760–768. PMID 22201612.
- Slanina T, Šebej P (June 2018). "Visible-light-activated photoCORMs: rational design of CO-releasing organic molecules absorbing in the tissue-transparent window". review article. Photochemical & Photobiological Sciences. 17 (6): 692–710. PMID 29796556.
