Enol
In
Keto–enol tautomerism refers to a
Enolization
- RC(=O)CHR′R′′ ⇌ RC(OH)=CR′R′′
In the case of ketones, the conversion is called a keto-enol tautomerism, although this name is often more generally applied to all such tautomerizations. Usually the equilibrium constant is so small that the enol is undetectable spectroscopically.
In some compounds with two (or more) carbonyls, the enol form becomes dominant. The behavior of
| carbonyl | enol | Kenolization |
|---|---|---|
| Acetaldehyde CH3CHO |
CH2=CHOH | 5.8×10−7 |
| Acetone CH3C(O)CH3 |
CH3C(OH)=CH2 | 5.12×10−7 |
| Methyl acetate CH3CO2CH3 |
CH2=CH(OH)OCH3 | 4×10−20 |
| Acetophenone C6H5C(O)CH3 |
C6H5C(OH)=CH2 | 1×10−8 |
| Acetylacetone CH3C(O)CH2C(O)CH3 |
CH3C(O)CH=C(OH)CH3 | 0.27 |
| Trifluoroacetylacetone CH3C(O)CH2C(O)CF3 |
CH3C(O)CH=C(OH)CF3 | 32 |
| Hexafluoroacetylacetone CF3C(O)CH2C(O)CF3 |
CF3C(O)CH=C(OH)CF3 | ~104 |
| Cyclohexa-2,4-dienone | Phenol C6H5OH |
>1012 |
Enols are derivatives of
The acid-catalyzed conversion of an enol to the keto form proceeds by proton transfer from O to carbon. The process does not occur intramolecularly, but requires participation of solvent or other mediators.
Stereochemistry of ketonization
If R1 and R2 (note equation at top of page) are different substituents, there is a new stereocenter formed at the alpha position when an enol converts to its keto form. Depending on the nature of the three R groups, the resulting products in this situation would be diastereomers or enantiomers.[citation needed]
Enediols
Enediols are alkenes with a hydroxyl group on each carbon of the C=C double bond. Normally such compounds are disfavored components in equilibria with
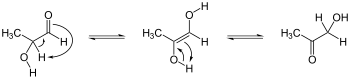
Keto-enediol tautomerizations. Enediol in the center; acyloin isomers at left and right. Ex. is hydroxyacetone, shown at right.
Phenols
Phenols represent a kind of enol. For some phenols and related compounds, the keto tautomer plays an important role. Many of the reactions of resorcinol involve the keto tautomer, for example. Naphthalene-1,4-diol exists in observable equilibrium with the diketone tetrahydronaphthalene-1,4-dione.[6]
Biochemistry
Keto–enol tautomerism is important in several areas of biochemistry.[citation needed]
The high phosphate-transfer potential of
The
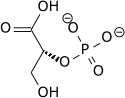
|
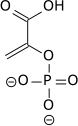
|

| ||||
| H2O | ADP | ATP | ||||

|

| |||||
| H2O | ||||||
Reactivity
Addition of electrophiles
The terminus of the double bond in enols is nucleophilic. Its reactions with electrophilic organic compounds is important in biochemistry as well as synthetic organic chemistry. In the former area, the fixation of carbon dioxide involves addition of CO2 to an enol.[citation needed]
Deprotonation: enolates
Deprotonation of enolizable ketones, aldehydes, and esters gives enolates.[8][9] Enolates can be trapped by the addition of electrophiles at oxygen. Silylation gives silyl enol ether.[10] Acylation gives esters such as vinyl acetate.[11]
Stable enols
In general, enols are less stable than their keto equivalents because of the favorability of the C=O double bond over C=C double bond. However, enols can be stabilized kinetically or thermodynamically.[citation needed]
Some enols are sufficiently stabilized kinetically so that they can be characterized.[citation needed]

Delocalization can stabilize the enol tautomer. Thus, very stable enols are phenols.[13] Another stabilizing factor in 1,3-dicarbonyls is intramolecular hydrogen bonding.[14] Both of these factors influence the enol-dione equilibrium in acetylacetone.
See also
- Alkenal
- Enolase
- Ketone
- Ynol
- Geminal diol, another form of ketones and aldehydes in water solutions
- Regioselectivity
References
- ^ ISBN 0-471-58589-0.
- ISBN 978-0-19-927029-3.
- .
- doi:10.1002/poc.3168.
- S2CID 260331550.
- PMID 16304647.
- ISBN 0-7167-3051-0.
- ISBN 978-0-471-72091-1
- ISBN 9783527671069.
- S2CID 241676899.
- .
- ^ Clayden, Jonathan (2012). Organic Chemistry. Oxford University Press. pp. 456–459.
- PMID 23320139.




