Nucleophilic conjugate addition
resonance structures that the β position is an electrophilic site which can react with a nucleophile. The negative charge in these structures is stored as an alkoxide anion. Such a nucleophilic addition is called a nucleophilic conjugate addition or 1,4-nucleophilic addition. The most important active alkenes are the aforementioned conjugated carbonyls and acrylonitriles
.
Reaction mechanism
Conjugate addition is the
keto-enol tautomerism to the saturated carbonyl compound. In vicinal difunctionalization
the proton is replaced by another electrophile.
Reactions
- Conjugated carbonyls react with secondary amines to form 3-aminocarbonyls (3-ketoamines). For example, the conjugate addition of methylamineto cyclohexen-2-one gives the compound 3-(N-methylamino)-cyclohexanone.
- Conjugated carbonyls react with diethylaluminum cyanide.
- The Gilman reagent is an effective nucleophile for 1,4-additions to conjugated carbonyls.
- The Michael reaction involves conjugate additions of enolatesto conjugated carbonyls.
- The Stork enamine reaction involves the conjugate addition of enaminesto conjugated carbonyls.
Scope
Conjugate addition is effective in the formation of new
methylvinylketone.[1]
![(4R',5R')-5-(5-Ethyl-2,2-dimethyl-[1,3]-dioxolan-4-yl)-pentan-2-one](http://upload.wikimedia.org/wikipedia/commons/7/7b/Conjugateadditionexample.png)
An example of an

In another example of asymmetric synthesis the α,β-unsaturated carbonyl compound first reacts with a
syn:anti ratio of 8:1 and 97% enantiomeric excess.[3]
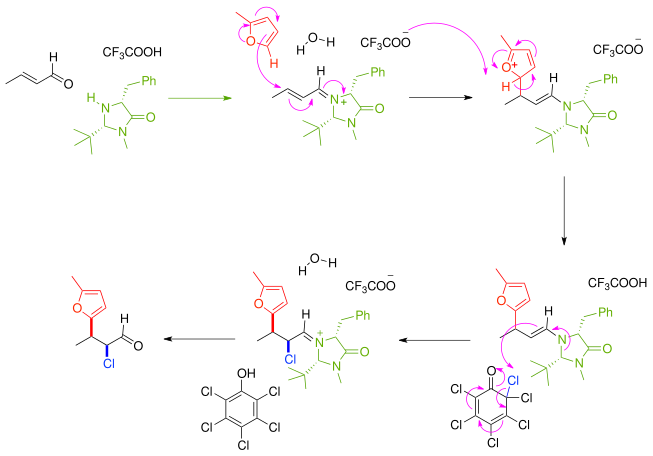
This principle is also applied in an enantioselective
domino conjugated addition of nucleophilic thiols such as benzylmercaptan and electrophilic DEAD.[4]

Toxicology
Suitably soluble Michael acceptors are toxic, because they alkylate DNA by conjugate addition. Such modification induces mutations, which are cytotoxic and carcinogenic. However, glutathione is also able to react with them and for example dimethyl fumarate reacts with it. [citation needed]
See also
References
- .
- ^ Tamio Hayashi; Makoto Takahashi; Yoshiaki Takaya; Masamichi Ogasawara (2004). "(R)-3-phenyl-cyclohexanone". Organic Syntheses; Collected Volumes, vol. 10, p. 609.
- .
- PMID 16277506.
