Diethyl azodicarboxylate

| |
| |
| Names | |
|---|---|
| IUPAC name
Diethyl diazenedicarboxylate
| |
| Systematic IUPAC name
Ethyl N-ethoxycarbonyliminocarbamate | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.016.202 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H10N2O4 | |
| Molar mass | 174.156 g·mol−1 |
| Appearance | Orange to red to orange liquid[2] |
| Density | 1.11 g/cm3[3] |
| Melting point | 6 °C (43 °F; 279 K)[4] |
| Boiling point | 104.5 °C (220.1 °F; 377.6 K) at 12 mm Hg[3] |
Refractive index (nD)
|
1.420 (20 °C)[3] |
| Hazards | |
| GHS labelling: | |
 
| |
| Danger | |
| H240, H302, H312, H315, H319, H332, H335 | |
| P210, P220, P234, P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P312, P304+P340, P305+P351+P338, P312, P321, P322, P330, P332+P313, P337+P313, P362, P363, P370+P378, P370+P380+P375, P403+P233, P403+P235, P405, P411, P420, P501 | |
| Flash point | 85 °C (185 °F; 358 K)[5] |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Diethyl azodicarboxylate, conventionally abbreviated as DEAD and sometimes as DEADCAT,[6][7] is an organic compound with the structural formula CH3CH2−O−C(=O)−N=N−C(=O)−O−CH2CH3. Its molecular structure consists of a central azo functional group, RN=NR, flanked by two ethyl ester groups. This orange-red liquid is a valuable reagent but also quite dangerous and explodes upon heating. Therefore, commercial shipment of pure diethyl azodicarboxylate is prohibited in the United States and is carried out either in solution or on polystyrene particles.
DEAD is an aza-
Properties
DEAD is an orange-red liquid which weakens its color to yellow or colorless upon dilution or chemical reaction. This color change is conventionally used for visual monitoring of the synthesis. DEAD dissolves in most common organic solvents, such as toluene, chloroform, ethanol,[9] tetrahydrofuran and dichloromethane[3][10] but has low solubility in water or carbon tetrachloride; the solubility in water is higher for the related azo compound dimethyl azodicarboxylate.[4]
DEAD is a strong electron acceptor and easily oxidizes a solution of
Preparation
Although available commercially, diethyl azodicarboxylate can be prepared fresh in the laboratory, especially if required in pure, non-diluted form. A two-step synthesis starts from hydrazine, first by alkylation with ethyl chloroformate, followed by treating the resulting diethyl hydrazodicarboxylate with chlorine (bubbling through the solution), hypochlorous acid, concentrated nitric acid or red fuming nitric acid. The reaction is carried out in an ice bath, and the reagents are added dropwise so that the temperature does not rise above 20 °C. Diethyl hydrazodicarboxylate is a solid with melting temperature of 131–133 °C which is collected as a residue; it is significantly more stable to heating than DEAD and is conventionally dried at a temperature of about 80 °C.[4][13]
Applications
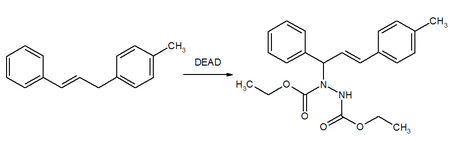
Mitsunobu reaction
DEAD is a reagent in the Mitsunobu reaction where it forms an adduct with phosphines (usually triphenylphosphine) and assists the synthesis of esters, ethers, amines and thioethers from alcohols. Reactions normally result in the inversion of molecular symmetry.
DEAD was used in the original 1967 article by Oyo Mitsunobu,[14] and his 1981 review on the use of diethyl azodicarboxylate is a top-cited chemistry article.[15][16] The Mitsunobu reaction has several applications in the synthesis of natural products and pharmaceuticals.
In the above reaction, which is assisted either by DEAD or
Michael reaction
The azo group in DEAD is a
The substitution of boronic acid esters proceeds similarly:[22]
Other reactions
DEAD is an efficient component in
DEAD can be used for synthesis of heterocyclic compounds. Thus, pyrazoline derivatives convert by condensation to α,β-unsaturated ketones:[25]
Another application is the use of DEAD as an enophile in ene reactions:
Safety
DEAD is toxic, shock and light sensitive; it can violently explode when its undiluted form is heated above 100 °C.
References
- ^ Diethyl azodiformate. Webbook.nist.gov (1972-07-28). Retrieved on 2011-03-15.
- ^ a b Safety (MSDS) data for diethyl azodicarboxylate Archived 2010-12-09 at the Wayback Machine. Msds.chem.ox.ac.uk. Retrieved on 2011-03-15.
- ^ ISBN 978-1-85617-567-8. Retrieved 12 March 2011.
- ^ a b c Kauer, J. C. "Diethyl azodicarboxylate". Organic Syntheses; Collected Volumes, vol. 4, p. 411.
- ^ L19348 Diethyl azodicarboxylate, 97% – Alfa Aesar – A Johnson Matthey Company. Alfa.com (1972-07-28). Retrieved on 2011-03-15.
- ISBN 978-1-4398-1164-1. Retrieved 12 March 2011.
- ISBN 978-0-471-08189-0. Retrieved 12 March 2011.
- OCLC 634820219.
- ^ ISSN 0022-3263.
- ^ Kelmara K. Kelly (2009) Novel isotope effects and organic reaction mechanisms, PhD thesis, Texas A&M University, p. 81
- .
- ISBN 978-0-471-93623-7.
- .
- .
- .
- ^ More than 4,300 citations by March 2011 according to Web of Science
- ^ Czernecki, Stanislas and Valery, Jean-marc "Process for preparing AZT (3'-azido-3'-deoxy-thymidine) and related compounds" U.S. patent 5,124,442 issued 23 June 1992
- .
- S2CID 93871095.
- PMID 7966151.
- PMID 15387609.
- PMID 16209627.
- ISSN 0078-6209.
- ^ Shi, Min; Zhao, Gui-Ling (2004). "Aza-Baylis–Hillman reactions of diisopropyl azodicarboxylate or diethyl azodicarboxylate with acrylates and acrylonitrile". .
- PMID 17286329.
- ISBN 978-0-19-855853-8. Retrieved 12 March 2011.
- ^ Diethyl Azodicarboxylate in Chemical Synthesis. Sigmaaldrich.com. Retrieved on 2011-03-15.
- ISBN 978-3-527-29823-5. Retrieved 12 March 2011.