Alkoxide
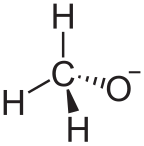

In
Structure
Alkali metal alkoxides are often oligomeric or polymeric compounds, especially when the R group is small (Me, Et).[3] [page needed] The alkoxide anion is a good bridging ligand, thus many alkoxides feature M2O or M3O linkages. In solution, the alkali metal derivatives exhibit strong ion-pairing, as expected for the alkali metal derivative of a strongly basic anion.

Preparation
From reducing metals
Alkoxides can be produced by several routes starting from an alcohol. Highly reducing metals react directly with alcohols to give the corresponding metal alkoxide. The alcohol serves as an acid, and hydrogen is produced as a by-product. A classic case is sodium methoxide produced by the addition of sodium metal to methanol:[citation needed]
- 2 CH3OH + 2 Na → 2 CH3ONa + H2
Other alkali metals can be used in place of sodium, and most alcohols can be used in place of methanol. Generally, the alcohol is used in excess and left to be used as a solvent in the reaction. Thus, an alcoholic solution of the alkali alkoxide is used. Another similar reaction occurs when an alcohol is reacted with a metal hydride such as NaH. The metal hydride removes the hydrogen atom from the hydroxyl group and forms a negatively charged alkoxide ion.
Properties
Reactions with alkyl halides
The alkoxide ion and its salts react with primary alkyl halides in an SN2 reaction to form an ether via the Williamson Ether Synthesis.[citation needed]
Hydrolysis and transesterification
Aliphatic metal alkoxides decompose in water as summarized in this idealized equation:
- Al(OR)3 + 3 H2O → Al(OH)3 + 3 ROH
In the
Formation of oxo-alkoxides
Many metal alkoxide compounds also feature oxo-
- RCO2R' + CH3O− → RCO2CH3 + R'OH
Thermal stability
Many metal alkoxides
Illustrative alkoxides
| name | molecular formula | comment |
|---|---|---|
| Tetraethyl orthosilicate | Si(OEt)4 | for sol-gel processing of Si oxides; Si(OMe)4 is avoided for safety reasons
|
| Aluminium isopropoxide | Al4(OiPr)12 | reagent for Meerwein–Ponndorf–Verley reduction |
| Potassium tert-butoxide, | K4(OtBu)4 | basic reagent in alcohol solution for organic elimination reactions |
Sodium methoxide
Sodium methoxide, also called
Sodium methoxide is produced on an industrial scale and available from a number of chemical companies.
Potassium methoxide
Potassium methoxide in alcoholic solution is commonly used as a catalyst for transesterification in the production of biodiesel.[7]
References
- )
- ISBN 9780136436690.
- ^ ISBN 978-0-08-048832-5.
- ISBN 9780792375210.
- PMID 24273149.
- ^ "Sodium Methoxide Material Safety Data Sheet (MSDS)". NOAA.gov. Archived from the original on 2009-02-18. Retrieved 2010-04-13.
- ISBN 978-1-893997-62-2
Further reading
- Turova, Nataliya Y. (2004). "Metal oxoalkoxides. Synthesis, properties and structures". S2CID 250920020.
