Phosphate-buffered saline
Phosphate-buffered saline (PBS) is a
Applications
PBS has many uses because it is isotonic and non-toxic to most cells. These uses include substance dilution and cell container rinsing. PBS with
Preparation
There are many different ways to prepare PBS solutions, common ones are Dulbecco's phosphate-buffered saline (DPBS)[2] and the Cold Spring Harbor protocol.[3] Some formulations of DPBS do not contain potassium and magnesium, while other ones contain calcium and/or magnesium (depending on whether or not the buffer is used on live or fixed tissue: the latter does not require CaCl2 or MgCl2 ).
| Salt | Concentration (mmol/L) | Concentration (g/L) |
|---|---|---|
| Na2HPO4 | 8.1 | 1.15 |
| KH2PO4 | 1.5 | 0.2 |
| NaCl | 137 | 8.0 |
| KCl | 2.7 | 0.2 |
| Salt | Concentration (mmol/L) | Concentration (g/L) |
|---|---|---|
| Na2HPO4 | 8.1 | 1.15 |
| KH2PO4 | 1.5 | 0.2 |
| NaCl | 137 | 8.0 |
| KCl | 2.7 | 0.2 |
| CaCl2 | 0.9 | 0.1 |
| MgCl2 | 0.5 | 0.1 |
| reagent | MW | mass (g) 10× | [mM] 10× | mass (g) 5× | [mM] 5× | mass (g) 1× | [mM] 1× |
|---|---|---|---|---|---|---|---|
| Na2HPO4 | 141.95897 | 14.1960 | 100 | 7.0980 | 0.0500 | 1.41960 | 10 |
| KH2PO4 | 136.08569 | 2.4496 | 18 | 1.2248 | 0.0090 | 0.24496 | 1.8 |
| NaCl | 58.44300 | 80.0669 | 1370 | 40.0335 | 0.6850 | 8.00669 | 137 |
| KCl | 74.55150 | 2.0129 | 27 | 1.0064 | 0.0135 | 0.20129 | 2.7 |
| pH = 7.4 |
Start with 800 mL of distilled water to dissolve all salts. Add distilled water to a total volume of 1 liter. The resultant 1× PBS will have a final concentration of 157 mM Na+, 140mM Cl−, 4.45mM K+, 10.1 mM HPO42−, 1.76 mM H2PO4− and a pH of 7.96. Add 2.84 mM of HCl to shift the buffer to 7.3 mM HPO42− and 4.6 mM H2PO4− for a final pH of 7.4 and a Cl− concentration of 142 mM.
The pH of PBS is ~7.4. When making buffer solutions, it is good practice to always measure the pH directly using a pH meter. If necessary, pH can be adjusted using hydrochloric acid or sodium hydroxide.
PBS can also be prepared by using commercially made PBS buffer tablets or pouches.[4]
If used in cell culturing, the solution can be dispensed into aliquots and sterilized by
Dependence of pH on ionic strength and temperature
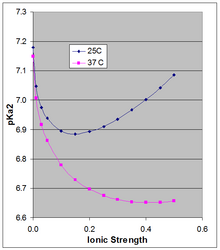
The Henderson–Hasselbach equation gives the pH of a solution relative to the pKa of the acid–base pair. However the pKa is dependent on ionic strength and temperature, and as it shifts so will the pH of a solution based on that acid–base pair. Because the doubly charged [HPO4]2− is stabilized more by high ionic strength than is the singly-charged [H2PO4]−, their pKa is somewhat dependent on ionic strength. The often-cited pKa of ~7.2 is the value extrapolated to zero ionic strength, and is not applicable at physiological ionic strength.
Phillips et al.
at 25 °C: pKa2 = 7.18 − 1.52 sqrt(µ) + 1.96 µ
at 37 °C: pKa2 = 7.15 − 1.56 sqrt(µ) + 1.22 µ
The pKa0 is weakly dependent on temperature. Phillips et al. reported ∆H0 at 25 °C of 760 cal/mol (3180 J/mol) and a linear dependence of pKa0 on 1/T (Van 't Hoff equation). The positive ∆H0 results in an increase in Ka, and thus a decrease in pKa0 with rising temperature, the change in pKa0 being 166 × the change in (1/T), which around 25 °C results in a change in pKa0 of −0.00187 per degree. This applies strictly to the extrapolated thermodynamic pKa0 at infinite dilution, and as the figure shows, the temperature effect can be much larger at higher ionic strength.
See also
References
- PMID 32414839.
- ^ PMID 13130792.
- ^ a b Phosphate-buffered saline (PBS) recipe. CSH Protocol
- ^ Phosphate buffered saline specification sheet. Medicago AB, (2010)
- PMID 14069537.
