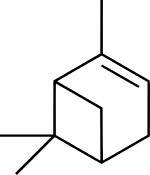
Pinene
| |
| Names | |
|---|---|
| IUPAC names
(1S,5S)-2,6,6-trimethylbicyclo[3.1.1]hept-2-ene
(1S,5S)-6,6-dimethyl-2-methylenebicyclo[3.1.1]heptane | |
| Identifiers | |
| |
3D model (
JSmol ) |
|
| ChEBI |
|
| ChemSpider | |
ECHA InfoCard
|
100.029.170 |
| EC Number |
|
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C10H16 | |
| Molar mass | 136.24 g/mol |
| Appearance | Liquid |
| Density | 0,86 g·cm−3 (alpha, 15 °C)[1][2] |
| Melting point | −62 to −55 °C (−80 to −67 °F; 211 to 218 K) (alpha)[1] |
| Boiling point | 155 to 156 °C (311 to 313 °F; 428 to 429 K) (alpha)[1] |
| Practically insoluble in water | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Pinene is a collection of unsaturated bicyclic
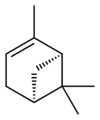
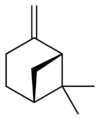
Isomers
| skeletal formula | 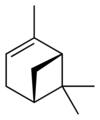 |
 |
 |

|
| perspective view | X | 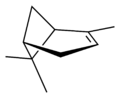 |
X | 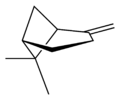
|
| ball-and-stick model | X | 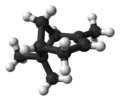 |
X | 
|
| name | (1R)-(+)-α-pinene | (1S)-(−)-α-pinene | (1R)-(+)-β-pinene | (1S)-(−)-β-pinene |
CAS number |
7785-70-8 | 7785-26-4 | 19902-08-0 | 18172-67-3 |
Biosynthesis
α-Pinene and β-pinene are both produced from geranyl pyrophosphate, via cyclisation of linaloyl pyrophosphate followed by loss of a proton from the carbocation equivalent. Researchers at the Georgia Institute of Technology and the Joint BioEnergy Institute have been able to synthetically produce pinene with a bacterium.[5]

Plants
Alpha-pinene appears in
Makrut lime fruit peel contains an essential oil comparable to lime fruit peel oil; its main components are limonene and β-pinene.[12]
The racemic mixture of the two forms of pinene is found in some oils like eucalyptus oil.[13]
Reactions
β-Pinene can be converted to α-pinene in the presence of strong bases.[14]
Selective oxidation of pinene occurs at the allylic position to give verbenone, along with pinene oxide, as well as verbenol and its hydroperoxide.[15][16]

Hydrogenation of pinene gives pinane, precursor to a useful pinanehydroperoxide.
The
Use
Pinenes, especially α, are the primary constituents of turpentine, a nature-derived solvent and fuel.[3]
The use of pinene as a biofuel in
References
- ^ a b c Record of alpha-Pinen in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 07-January-2016.
- ^ Record of beta-Pinen in the GESTIS Substance Database of the Institute for Occupational Safety and Health, accessed on 07-January-2016.
- ^ ISBN 978-3527306732.
- .
- ^ PMID 24679043.

- ISBN 9780429155666.
- PMID 19729299.
- ^ PMID 21749363.
- PMID 20670669.
- PMID 20015509.
- .
- ^ Kasuan N (2013). "Extraction of Citrus hystrix D.C. (Kaffir Lime) Essential Oil Using Automated Steam Distillation Process: Analysis of Volatile Compounds" (PDF). Malaysian Journal of Analytical Sciences. 17 (3): 359–369.
- ^ "alpha-Pinene - Compound Summary". PubChem. NCBI. Retrieved 14 Nov 2017.
- .
- PMID 20017184.
- doi:10.15227/orgsyn.072.0057.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- .
Bibliography
- Mann, J.; Davidson, R. S.; Hobbs, J. B.; Banthorpe, D. V.; Harborne, J. B. (1994). Natural Products. Harlow, UK: Addison Wesley Longman Ltd. pp. 309–311. ISBN 978-0-582-06009-8.
