Polymer
| Polymer science |
|---|
 |

A polymer (/ˈpɒlɪmər/[2][3]) is a
Polymers are studied in the fields of

Etymology
The term "polymer" derives from
Common examples

Polymers are of two types: naturally occurring and synthetic or man made.
Natural
Space polymer
Hemoglycin (previously termed hemolithin) is a space polymer that is the first polymer of amino acids found in meteorites.[12][13][14]
Synthetic
The list of synthetic polymers, roughly in order of worldwide demand, includes polyethylene, polypropylene, polystyrene, polyvinyl chloride, synthetic rubber, phenol formaldehyde resin (or Bakelite), neoprene, nylon, polyacrylonitrile, PVB, silicone, and many more. More than 330 million tons of these polymers are made every year (2015).[15]
Most commonly, the continuously linked backbone of a polymer used for the preparation of plastics consists mainly of carbon atoms. A simple example is polyethylene ('polythene' in British English), whose repeat unit or monomer is ethylene. Many other structures do exist; for example, elements such as silicon form familiar materials such as silicones, examples being Silly Putty and waterproof plumbing sealant. Oxygen is also commonly present in polymer backbones, such as those of polyethylene glycol, polysaccharides (in glycosidic bonds), and DNA (in phosphodiester bonds).
Synthesis

Polymerization is the process of combining many small molecules known as monomers into a covalently bonded chain or network. During the polymerization process, some chemical groups may be lost from each monomer. This happens in the polymerization of PET polyester. The monomers are terephthalic acid (HOOC—C6H4—COOH) and ethylene glycol (HO—CH2—CH2—OH) but the repeating unit is —OC—C6H4—COO—CH2—CH2—O—, which corresponds to the combination of the two monomers with the loss of two water molecules. The distinct piece of each monomer that is incorporated into the polymer is known as a repeat unit or monomer residue.
Synthetic methods are generally divided into two categories,

Newer methods, such as plasma polymerization do not fit neatly into either category. Synthetic polymerization reactions may be carried out with or without a catalyst. Laboratory synthesis of biopolymers, especially of proteins, is an area of intensive research.
Biological synthesis
There are three main classes of biopolymers:
Modification of natural polymers
Naturally occurring polymers such as
Structure
The structure of a polymeric material can be described at different length scales, from the sub-nm length scale up to the macroscopic one. There is in fact a hierarchy of structures, in which each stage provides the foundations for the next one.[19] The starting point for the description of the structure of a polymer is the identity of its constituent monomers. Next, the microstructure essentially describes the arrangement of these monomers within the polymer at the scale of a single chain. The microstructure determines the possibility for the polymer to form phases with different arrangements, for example through crystallization, the glass transition or microphase separation.[20] These features play a major role in determining the physical and chemical properties of a polymer.
Monomers and repeat units
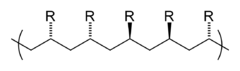
The identity of the repeat units (monomer residues, also known as "mers") comprising a polymer is its first and most important attribute. Polymer nomenclature is generally based upon the type of monomer residues comprising the polymer. A polymer which contains only a single type of repeat unit is known as a homopolymer, while a polymer containing two or more types of repeat units is known as a copolymer.[21] A terpolymer is a copolymer which contains three types of repeat units.[22]
Polystyrene is composed only of styrene-based repeat units, and is classified as a homopolymer. Polyethylene terephthalate, even though produced from two different monomers (ethylene glycol and terephthalic acid), is usually regarded as a homopolymer because only one type of repeat unit is formed. Ethylene-vinyl acetate contains more than one variety of repeat unit and is a copolymer. Some biological polymers are composed of a variety of different but structurally related monomer residues; for example, polynucleotides such as DNA are composed of four types of nucleotide subunits.
Homopolymers and copolymers (examples) 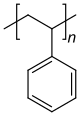
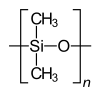
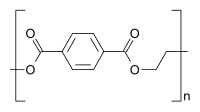
Homopolymer polystyrene Homopolymer polydimethylsiloxane, a silicone. The main chain is formed of silicon and oxygen atoms. The homopolymer polyethylene terephthalate has only one repeat unit. Copolymer 1,3-butadieneform two repeating units, which can alternate in any order in the macromolecule, making the polymer thus a random copolymer.
A polymer containing ionizable subunits (e.g., pendant carboxylic groups) is known as a polyelectrolyte or ionomer, when the fraction of ionizable units is large or small respectively.
Microstructure
The microstructure of a polymer (sometimes called configuration) relates to the physical arrangement of monomer residues along the backbone of the chain.
While branched and unbranched polymers are usually thermoplastics, many

Linear, unbranched macromolecule
Branched macromolecule
Semi-crystalline structure of an unbranched polymer
Slightly cross-linked polymer (elastomer)
Highly cross-linked polymer (thermoset)
Polymer architecture

An important microstructural feature of a polymer is its architecture and shape, which relates to the way branch points lead to a deviation from a simple linear chain.
Chain length
A common means of expressing the length of a chain is the
The physical properties[30] of polymer strongly depend on the length (or equivalently, the molecular weight) of the polymer chain.[31] One important example of the physical consequences of the molecular weight is the scaling of the viscosity (resistance to flow) in the melt.[32] The influence of the weight-average molecular weight () on the melt viscosity () depends on whether the polymer is above or below the onset of entanglements. Below the entanglement molecular weight[clarification needed], , whereas above the entanglement molecular weight, . In the latter case, increasing the polymer chain length 10-fold would increase the viscosity over 1000 times.[33][page needed] Increasing chain length furthermore tends to decrease chain mobility, increase strength and toughness, and increase the glass-transition temperature (Tg).[34] This is a result of the increase in chain interactions such as van der Waals attractions and entanglements that come with increased chain length.[35][36] These interactions tend to fix the individual chains more strongly in position and resist deformations and matrix breakup, both at higher stresses and higher temperatures.
Monomer arrangement in copolymers
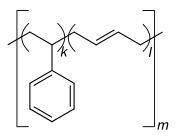
Copolymers are classified either as statistical copolymers, alternating copolymers, block copolymers, graft copolymers or gradient copolymers. In the schematic figure below, Ⓐ and Ⓑ symbolize the two repeat units.
- Alternating copolymers possess two regularly alternating monomer residues:[37] (AB)
n. An example is the equimolar copolymer of styrene and maleic anhydride formed by free-radical chain-growth polymerization.[38] A step-growth copolymer such as Nylon 66 can also be considered a strictly alternating copolymer of diamine and diacid residues, but is often described as a homopolymer with the dimeric residue of one amine and one acid as a repeat unit.[39] - Periodic copolymers have more than two species of monomer units in a regular sequence.[40]
- Statistical copolymers have monomer residues arranged according to a statistical rule. A statistical copolymer in which the probability of finding a particular type of monomer residue at a particular point in the chain is independent of the types of surrounding monomer residue may be referred to as a truly random copolymer.[41][42] For example, the chain-growth copolymer of vinyl chloride and vinyl acetate is random.[38]
- Block copolymers have long sequences of different monomer units.[38][39] Polymers with two or three blocks of two distinct chemical species (e.g., A and B) are called diblock copolymers and triblock copolymers, respectively. Polymers with three blocks, each of a different chemical species (e.g., A, B, and C) are termed triblock terpolymers.
- Graft or grafted copolymers contain side chains or branches whose repeat units have a different composition or configuration than the main chain.[39] The branches are added on to a preformed main chain macromolecule.[38]
Monomers within a copolymer may be organized along the backbone in a variety of ways. A copolymer containing a controlled arrangement of monomers is called a sequence-controlled polymer.[43] Alternating, periodic and block copolymers are simple examples of sequence-controlled polymers.
Tacticity
Tacticity describes the relative
Morphology
Polymer morphology generally describes the arrangement and microscale ordering of polymer chains in space. The macroscopic physical properties of a polymer are related to the interactions between the polymer chains.
 Randomly oriented polymer |
 Interlocking of several polymers |
- Disordered polymers: In the solid state, atactic polymers, polymers with a high degree of conformations of the molecules are frozen. Hooking and entanglement of chain molecules lead to a "mechanical bond" between the chains. Intermolecularand intramolecular attractive forces only occur at sites where molecule segments are close enough to each other. The irregular structures of the molecules prevent a narrower arrangement.
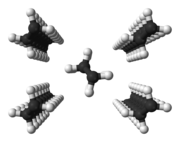 Polyethylene: zigzag conformation of molecules in close packed chains |
 Lamella with tie molecules |
 Spherulite |
 polypropylene helix |
p-Aramid , red dotted: hydrogen bonds
|
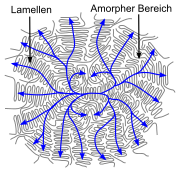
- Linear polymers with periodic structure, low branching and stereoregularity (e. g. not atactic) have a semi-crystalline structure in the solid state.[44] In simple polymers (such as polyethylene), the chains are present in the crystal in zigzag conformation. Several zigzag conformations form dense chain packs, called crystallites or lamellae. The lamellae are much thinner than the polymers are long (often about 10 nm).[45] They are formed by more or less regular folding of one or more molecular chains. Amorphous structures exist between the lamellae. Individual molecules can lead to entanglements between the lamellae and can also be involved in the formation of two (or more) lamellae (chains than called tie molecules). Several lamellae form a superstructure, a spherulite, often with a diameter in the range of 0.05 to 1 mm.[45]
- The type and arrangement of (functional) residues of the repeat units effects or determines the crystallinity and strength of the secondary valence bonds. In isotactic polypropylene, the molecules form a helix. Like the zigzag conformation, such helices allow a dense chain packing. Particularly strong intermolecular interactions occur when the residues of the repeating units allow the formation of hydrogen bonds, as in the case of p-aramid. The formation of strong intramolecular associations may produce diverse folded states of single linear chains with distinct circuit topology. Crystallinity and superstructure are always dependent on the conditions of their formation, see also: crystallization of polymers. Compared to amorphous structures, semi-crystalline structures lead to a higher stiffness, density, melting temperature and higher resistance of a polymer.
- Cross-linked polymers: Wide-meshed cross-linked polymers are elastomers and cannot be molten (unlike thermoplastics); heating cross-linked polymers only leads to decomposition. Thermoplastic elastomers, on the other hand, are reversibly "physically crosslinked" and can be molten. Block copolymers in which a hard segment of the polymer has a tendency to crystallize and a soft segment has an amorphous structure are one type of thermoplastic elastomers: the hard segments ensure wide-meshed, physical crosslinking.
 Wide-meshed cross-linked polymer (elastomer) |
tensile stress
|
 Crystallites as "crosslinking sites": one type of thermoplastic elastomer |
 Semi-crystalline thermoplastic elastomer under tensile stress |
Crystallinity
When applied to polymers, the term crystalline has a somewhat ambiguous usage. In some cases, the term crystalline finds identical usage to that used in conventional crystallography. For example, the structure of a crystalline protein or polynucleotide, such as a sample prepared for x-ray crystallography, may be defined in terms of a conventional unit cell composed of one or more polymer molecules with cell dimensions of hundreds of angstroms or more. A synthetic polymer may be loosely described as crystalline if it contains regions of three-dimensional ordering on atomic (rather than macromolecular) length scales, usually arising from intramolecular folding or stacking of adjacent chains. Synthetic polymers may consist of both crystalline and amorphous regions; the degree of crystallinity may be expressed in terms of a weight fraction or volume fraction of crystalline material. Few synthetic polymers are entirely crystalline.[46] The crystallinity of polymers is characterized by their degree of crystallinity, ranging from zero for a completely non-crystalline polymer to one for a theoretical completely crystalline polymer. Polymers with microcrystalline regions are generally tougher (can be bent more without breaking) and more impact-resistant than totally amorphous polymers.[47] Polymers with a degree of crystallinity approaching zero or one will tend to be transparent, while polymers with intermediate degrees of crystallinity will tend to be opaque due to light scattering by crystalline or glassy regions. For many polymers, crystallinity may also be associated with decreased transparency.
Chain conformation
The space occupied by a polymer molecule is generally expressed in terms of radius of gyration, which is an average distance from the center of mass of the chain to the chain itself. Alternatively, it may be expressed in terms of pervaded volume, which is the volume spanned by the polymer chain and scales with the cube of the radius of gyration.[48] The simplest theoretical models for polymers in the molten, amorphous state are ideal chains.
Properties
Polymer properties depend of their structure and they are divided into classes according to their physical bases. Many physical and chemical properties describe how a polymer behaves as a continuous macroscopic material. They are classified as bulk properties, or intensive properties according to thermodynamics.
Mechanical properties

The bulk properties of a polymer are those most often of end-use interest. These are the properties that dictate how the polymer actually behaves on a macroscopic scale.
Tensile strength
The
Young's modulus of elasticity
Transport properties
Transport properties such as diffusivity describe how rapidly molecules move through the polymer matrix. These are very important in many applications of polymers for films and membranes.
The movement of individual macromolecules occurs by a process called reptation in which each chain molecule is constrained by entanglements with neighboring chains to move within a virtual tube. The theory of reptation can explain polymer molecule dynamics and viscoelasticity.[51]
Phase behavior
Crystallization and melting
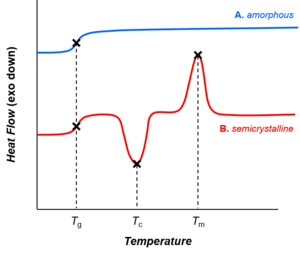
Depending on their chemical structures, polymers may be either semi-crystalline or amorphous. Semi-crystalline polymers can undergo crystallization and melting transitions, whereas amorphous polymers do not. In polymers, crystallization and melting do not suggest solid-liquid phase transitions, as in the case of water or other molecular fluids. Instead, crystallization and melting refer to the phase transitions between two solid states (i.e., semi-crystalline and amorphous). Crystallization occurs above the glass-transition temperature (Tg) and below the melting temperature (Tm).
Glass transition
All polymers (amorphous or semi-crystalline) go through glass transitions. The glass-transition temperature (Tg) is a crucial physical parameter for polymer manufacturing, processing, and use. Below Tg, molecular motions are frozen and polymers are brittle and glassy. Above Tg, molecular motions are activated and polymers are rubbery and viscous. The glass-transition temperature may be engineered by altering the degree of branching or crosslinking in the polymer or by the addition of plasticizers.[52]
Whereas crystallization and melting are first-order phase transitions, the glass transition is not.[53] The glass transition shares features of second-order phase transitions (such as discontinuity in the heat capacity, as shown in the figure), but it is generally not considered a thermodynamic transition between equilibrium states.
Mixing behavior

In general, polymeric mixtures are far less
Furthermore, the phase behavior of polymer solutions and mixtures is more complex than that of small molecule mixtures. Whereas most small molecule solutions exhibit only an upper critical solution temperature phase transition (UCST), at which phase separation occurs with cooling, polymer mixtures commonly exhibit a lower critical solution temperature phase transition (LCST), at which phase separation occurs with heating.
In dilute solutions, the properties of the polymer are characterized by the interaction between the solvent and the polymer. In a good solvent, the polymer appears swollen and occupies a large volume. In this scenario, intermolecular forces between the solvent and monomer subunits dominate over intramolecular interactions. In a bad solvent or poor solvent, intramolecular forces dominate and the chain contracts. In the
Inclusion of plasticizers
Inclusion of plasticizers tends to lower Tg and increase polymer flexibility. Addition of the plasticizer will also modify dependence of the glass-transition temperature Tg on the cooling rate.[54] The mobility of the chain can further change if the molecules of plasticizer give rise to hydrogen bonding formation. Plasticizers are generally small molecules that are chemically similar to the polymer and create gaps between polymer chains for greater mobility and fewer interchain interactions. A good example of the action of plasticizers is related to polyvinylchlorides or PVCs. A uPVC, or unplasticized polyvinylchloride, is used for things such as pipes. A pipe has no plasticizers in it, because it needs to remain strong and heat-resistant. Plasticized PVC is used in clothing for a flexible quality. Plasticizers are also put in some types of cling film to make the polymer more flexible.
Chemical properties
The attractive forces between polymer chains play a large part in determining the polymer's properties. Because polymer chains are so long, they have many such interchain interactions per molecule, amplifying the effect of these interactions on the polymer properties in comparison to attractions between conventional molecules. Different side groups on the polymer can lend the polymer to
The intermolecular forces in polymers can be affected by
When a polymer is dispersed or dissolved in a liquid, such as in commercial products like paints and glues, the chemical properties and molecular interactions influence how the solution flows and can even lead to
Optical properties
Polymers such as PMMA and HEMA:MMA are used as matrices in the gain medium of solid-state dye lasers, also known as solid-state dye-doped polymer lasers. These polymers have a high surface quality and are also highly transparent so that the laser properties are dominated by the laser dye used to dope the polymer matrix. These type of lasers, that also belong to the class of organic lasers, are known to yield very narrow linewidths which is useful for spectroscopy and analytical applications.[55] An important optical parameter in the polymer used in laser applications is the change in refractive index with temperature also known as dn/dT. For the polymers mentioned here the (dn/dT) ~ −1.4 × 10−4 in units of K−1 in the 297 ≤ T ≤ 337 K range.[56]
Electrical properties
Most conventional polymers such as polyethylene are electrical insulators, but the development of polymers containing π-conjugated bonds has led to a wealth of polymer-based semiconductors, such as polythiophenes. This has led to many applications in the field of organic electronics.
Applications
Nowadays, synthetic polymers are used in almost all walks of life. Modern society would look very different without them. The spreading of polymer use is connected to their unique properties: low density, low cost, good thermal/electrical insulation properties, high resistance to corrosion, low-energy demanding polymer manufacture and facile processing into final products. For a given application, the properties of a polymer can be tuned or enhanced by combination with other materials, as in composites. Their application allows to save energy (lighter cars and planes, thermally insulated buildings), protect food and drinking water (packaging), save land and lower use of fertilizers (synthetic fibres), preserve other materials (coatings), protect and save lives (hygiene, medical applications). A representative, non-exhaustive list of applications is given below.
- Clothing, sportswear and accessories: polyester and PVC clothing, spandex, sport shoes, wetsuits, footballs and billiard balls, skis and snowboards, rackets, parachutes, sails, tents and shelters.
- Electronic and photonic technologies: organic field effect transistors (OFET), light emitting diodes (OLED) and solar cells, television components, compact discs (CD), photoresists, holography.
- Packaging and containers: films, bottles, food packaging, barrels.
- Insulation: electrical and thermal insulation, spray foams.
- Construction and structural applications: garden furniture, PVC windows, flooring, sealing, pipes.
- Paints, glues and lubricants: varnish, adhesives, dispersants, anti-graffiti coatings, antifouling coatings, non-stick surfaces, lubricants.
- Car parts: tires, bumpers, windshields, windscreen wipers, fuel tanks, car seats.
- Household items: Rubik's cube).
- Medical applications: blood bag, syringes, rubber gloves, surgical suture, contact lenses, prosthesis, controlled drug delivery and release, matrices for cell growth.
- Personal hygiene and healthcare: diapers using superabsorbent polymers, toothbrushes, cosmetics, shampoo, condoms.
- Security: personal protective equipment, bulletproof vests, space suits, ropes.
- Separation technologies: synthetic membranes, fuel cell membranes, filtration, ion-exchange resins.
- Money: polymer banknotes and payment cards.
- 3D printing.
Standardized nomenclature
There are multiple conventions for naming polymer substances. Many commonly used polymers, such as those found in consumer products, are referred to by a common or trivial name. The trivial name is assigned based on historical precedent or popular usage rather than a standardized naming convention. Both the
| Common name | ACS name | IUPAC name |
|---|---|---|
Poly(ethylene oxide) or PEO
|
Poly(oxyethylene) | Poly(oxyethylene) |
Poly(ethylene terephthalate) or PET
|
Poly(oxy-1,2-ethanediyloxycarbonyl-1,4-phenylenecarbonyl) | Poly(oxyethyleneoxyterephthaloyl) |
| Nylon 6 or Polyamide 6 | Poly[imino(1-oxo-1,6-hexanediyl)] | Poly[azanediyl(1-oxohexane-1,6-diyl)] |
In both standardized conventions, the polymers' names are intended to reflect the monomer(s) from which they are synthesized (source based nomenclature) rather than the precise nature of the repeating subunit. For example, the polymer synthesized from the simple alkene
However, IUPAC structure based nomenclature is based on naming of the preferred constitutional repeating unit.[60]
Characterization
Polymer characterization spans many techniques for determining the chemical composition, molecular weight distribution, and physical properties. Select common techniques include the following:
- dispersity.
- Scattering techniques, such as block polymers, polymeric micelles, and other materials.
- Wide-angle X-ray scattering (also called wide-angle X-ray diffraction) is used to determine the crystalline structure of polymers (or lack thereof).
- Spectroscopy techniques, including Fourier-transform infrared spectroscopy, Raman spectroscopy, and nuclear magnetic resonance spectroscopy, can be used to determine the chemical composition.
- Differential scanning calorimetry is used to characterize the thermal properties of polymers, such as the glass-transition temperature, crystallization temperature, and melting temperature. The glass-transition temperature can also be determined by dynamic mechanical analysis.
- Thermogravimetryis a useful technique to evaluate the thermal stability of the polymer.
- Rheology is used to characterize the flow and deformation behavior. It can be used to determine the viscosity, modulus, and other rheological properties. Rheology is also often used to determine the molecular architecture (molecular weight, molecular weight distribution, branching) and to understand how the polymer can be processed.
Degradation

Polymer degradation is a change in the properties—tensile strength,
Although such changes are frequently undesirable, in some cases, such as
The susceptibility of a polymer to degradation depends on its structure. Epoxies and chains containing aromatic functionalities are especially susceptible to
The degradation of polyethylene occurs by random scission—a random breakage of the bonds that hold the atoms of the polymer together. When heated above 450 °C, polyethylene degrades to form a mixture of hydrocarbons. In the case of chain-end scission, monomers are released and this process is referred to as unzipping or depolymerization. Which mechanism dominates will depend on the type of polymer and temperature; in general, polymers with no or a single small substituent in the repeat unit will decompose via random-chain scission.
The sorting of polymer waste for recycling purposes may be facilitated by the use of the resin identification codes developed by the Society of the Plastics Industry to identify the type of plastic.
Product failure

Failure of

Polymer oxidation has caused accidents involving medical devices. One of the oldest known failure modes is ozone cracking caused by chain scission when ozone gas attacks susceptible elastomers, such as natural rubber and nitrile rubber. They possess double bonds in their repeat units which are cleaved during ozonolysis. Cracks in fuel lines can penetrate the bore of the tube and cause fuel leakage. If cracking occurs in the engine compartment, electric sparks can ignite the gasoline and can cause a serious fire. In medical use degradation of polymers can lead to changes of physical and chemical characteristics of implantable devices.[61]
History
Polymers have been essential components of commodities since the early days of humankind. The use of wool (keratin), cotton and linen fibres (cellulose) for garments, paper reed (cellulose) for paper are just a few examples of how ancient societies exploited polymer-containing raw materials to obtain artefacts. The latex sap of "caoutchouc" trees (natural rubber) reached Europe in the 16th century from South America long after the Olmec, Maya and Aztec had started using it as a material to make balls, waterproof textiles and containers.[62]
The chemical manipulation of polymers dates back to the 19th century, although at the time the nature of these species was not understood. The behaviour of polymers was initially rationalised according to the theory proposed by Thomas Graham which considered them as colloidal aggregates of small molecules held together by unknown forces.
Notwithstanding the lack of theoretical knowledge, the potential of polymers to provide innovative, accessible and cheap materials was immediately grasped. The work carried out by
In 1920, Hermann Staudinger published his seminal work "Über Polymerisation",[64] in which he proposed that polymers were in fact long chains of atoms linked by covalent bonds. His work was debated at length, but eventually it was accepted by the scientific community. Because of this work, Staudinger was awarded the Nobel Prize in 1953.[65]
After the 1930s polymers entered a golden age during which new types were discovered and quickly given commercial applications, replacing naturally-sourced materials. This development was fuelled by an industrial sector with a strong economic drive and it was supported by a broad academic community that contributed innovative syntheses of monomers from cheaper raw material, more efficient polymerisation processes, improved techniques for polymer characterisation and advanced, theoretical understanding of polymers.[63]

Since 1953, six Nobel prizes have been awarded in the area of polymer science, excluding those for research on biological macromolecules. This further testifies to its impact on modern science and technology. As Lord Todd summarised in 1980, "I am inclined to think that the development of polymerization is perhaps the biggest thing that chemistry has done, where it has had the biggest effect on everyday life".[67]
See also
- Ideal chain
- Catenation
- Inorganic polymer
- Important publications in polymer chemistry
- Oligomer
- Polymer adsorption
- Polymer classes
- Polymer engineering
- Polymery (botany)
- Reactive compatibilization
- Sequence-controlled polymer
- Shape-memory polymer
- Sol–gel process
- Supramolecular polymer
- Thermoplastic
- Thermosetting polymer
References
- PMID 16277495.
- The Free Dictionary. Retrieved 23 July 2013.
- ^ "Define polymer". Dictionary Reference. Retrieved 23 July 2013.
- ^ "Polymer on Britannica". 25 December 2023.
- ISBN 978-1-56676-559-6.
- ISBN 978-0-19-856526-0.
- ^ Saberi, A.; Bakhsheshi-Rad, H.R.; Abazari, S.; Ismail, A.F.; Sharif, S.; Ramakrishna, S.; Daroonparvar, M.; Berto, F. A Comprehensive Review on Surface Modifications of Biodegradable Magnesium-Based Implant Alloy: Polymer Coatings Opportunities and Challenges. Coatings 2021, 11, 747. https://doi.org/10.3390/coatings11070747
- ^ If two substances had molecular formulae such that one was an integer multiple of the other – e.g., acetylene (C2H2) and benzene (C6H6) – Berzelius called the multiple formula "polymeric". See: Jöns Jakob Berzelius (1833) "Isomerie, Unterscheidung von damit analogen Verhältnissen" (Isomeric, distinction from relations analogous to it), Jahres-Bericht über die Fortschitte der physischen Wissenschaften …, 12: 63–67. From page 64: "Um diese Art von Gleichheit in der Zusammensetzung, bei Ungleichheit in den Eigenschaften, bezeichnen zu können, möchte ich für diese Körper die Benennung polymerische (von πολυς mehrere) vorschlagen." (In order to be able to denote this type of similarity in composition [which is accompanied] by differences in properties, I would like to propose the designation "polymeric" (from πολυς, several) for these substances.)
Originally published in 1832 in Swedish as: Jöns Jacob Berzelius (1832) "Isomeri, dess distinktion från dermed analoga förhållanden," Årsberättelse om Framstegen i Fysik och Kemi, pages 65–70; the word "polymeriska" appears on page 66. - doi:10.1021/ed085p624. Archived from the original(PDF) on 18 June 2018. Retrieved 4 March 2013.
- .
- ISBN 978-0-13-065056-6.
- S2CID 97089690.
- PMID 36171277.
- ^ Staff (29 June 2021). "Polymers in meteorites provide clues to early solar system". Science Digest. Retrieved 9 January 2023.
- ^ "World Plastics Production" (PDF).
- ISBN 978-0-471-70606-9.
- ^ Sperling, p. 11
- ^ Sperling, p. 15
- ^ Sperling, p. 29
- ISBN 9780511801280.
- ^ Rudin, p. 17
- ^ Cowie, p. 4
- ^ Sperling, p. 30
- ^ ISBN 978-0-19-852059-7.
- ^ McCrum, p. 30
- ^ Rubinstein, p. 3
- ^ McCrum, p. 33
- ^ Rubinstein, pp. 23–24
- ^ Painter, p. 22
- ISBN 978-0-8014-1203-5.
- ^ Rubinstein, p. 5
- ^ McCrum, p. 37
- ^ Introduction to Polymer Science and Chemistry: A Problem-Solving Approach By Manas Chanda
- .
- ISBN 978-90-481-2230-1.
- .
- ^ Painter, p. 14
- ^ a b c d Rudin, p. 18–20
- ^ a b c Cowie, p. 104
- . Retrieved 9 April 2020.
- ^ Painter, p. 15
- ^ Sperling, p. 47
- S2CID 206549042.
- ^ a b Bernd Tieke: Makromolekulare Chemie. 3. Auflage, Wiley-VCH, Weinheim 2014, S. 295f (in German).
- ^ a b Wolfgang Kaiser: Kunststoffchemie für Ingenieure. 3. Auflage, Carl Hanser, München 2011, S. 84.
- ^ Sayed, Abu (August 2014). "Types of polymer: Requirements of fibre forming polymer". Textile Apex.
- ISBN 978-0-13-065056-6.
- ^ Rubinstein, p. 13
- ISBN 978-0-7506-2766-5.
- ISBN 978-0-521-86675-0. Archived from the originalon 2 November 2013. Retrieved 31 December 2018.
- ISBN 0-13-018168-4.
- ISBN 978-0-471-47936-9.
- S2CID 98823962. Retrieved 31 December 2018.
- S2CID 219911779
- PMID 18324163.
- ISBN 978-0122226960.
- ^ CAS: Index Guide, Appendix IV ((c) 1998)
- .
- ^ Wilks, E.S. "Macromolecular Nomenclature Note No. 18". Archived from the original on 25 September 2003.
- S2CID 95629051.
- PMID 26315946.
- ISSN 0022-233X.
- ^ S2CID 219539020.
- ISSN 0365-9488.
- ^ "The Nobel Prize in Chemistry 1953". NobelPrize.org. Retrieved 25 June 2020.
- S2CID 219539020.
- ISSN 0009-2347.
Bibliography
- Cowie, J. M. G. (John McKenzie Grant) (1991). Polymers: chemistry and physics of modern materials. Glasgow: Blackie. ISBN 978-0-412-03121-2.
- Hall, Christopher (1989). Polymer materials (PDF) (2nd ed.). London; New York: Macmillan. ISBN 978-0-333-46379-6.
- Rudin, Alfred (1982). The elements of polymer science and engineering. Academic Press. ISBN 978-0-12-601680-2.
- Wright, David C. (2001). Environmental Stress Cracking of Plastics. RAPRA. ISBN 978-1-85957-064-7.
External links
This article's use of external links may not follow Wikipedia's policies or guidelines. (June 2021) |