Polyaspartic acid
| |
| Names | |
|---|---|
| Other names
PASP
| |
| Identifiers | |
| |
| ChemSpider |
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| (C4H5NO3)n | |
| Molar mass | variable |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |

Polyaspartic acid (PASA) is a
Sodium polyaspartate is a sodium salt of polyaspartic acid.
In nature, PASA has been found in as fragments of larger
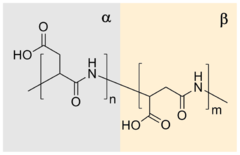
Properties and structure
Due to presence of
Synthesis


Many different routes lead to PASA. In the simplest[10] and the oldest approach[6] aspartic acid is heated to induce dehydration. In a subsequent step the resulting polysuccinimide is treated with aqueous sodium hydroxide, which yields partial opening of the succinimide rings. In this process sodium-DL-(α,β)-poly(aspartate) with 30% α-linkages and 70% β-linkages[11] randomly distributed along the polymer chain,[12] and racemized chiral center of aspartic acid is produced.[13] There were many catalysts reported for improving thermal polymerization method. Main benefits from their application is increasing of the conversion rate and higher molecular weight of the product.[14][15] Polyaspartic acid can also be synthesized by polymerization of
High control over repeating unit isomers can be achieved by polymerization of N-carboxyanhydride (NCA) derivatives,[17] by polymerization of aspartic acid esters[18] or by application of enzyme catalyzed reaction.[19] Pure homopolymers, D- or L-PASA with α- or β-links only, can be synthesized using those methods.The

Applications
Polyaspartic acid and its derivatives are biodegradable alternatives to traditional polyanionic materials, in particular polyacrylic acid.[20] PASA has ability to inhibit deposition of calcium carbonate, calcium sulfate, barium sulfate, and calcium phosphate and can be used as an antiscaling agent in cooling water systems, water desalination processes, and waste water treatment operations.[21] In addition and due to its ability to chelate metal ions, it provides corrosion inhibition.[11] It can also be used as biodegradable detergent and dispersant for various applications.[22]
PASA also has a variety of
As it can be synthesized in an environmentally friendly way and is
See also
References
- ^ ISBN 978-3527306732.
- ^ S2CID 232761877.
- .
- ^ PMID 31799235.
- ^ ISBN 978-0-8412-1886-4.
- ^ ISBN 978-3-527-30222-2.
- .
- S2CID 40153017.
- S2CID 33648417.
- .
- ^ ISBN 978-0-8412-3133-7.
- S2CID 85201969.
- .
- .
- S2CID 85163135.
- ^ US patent 5468838, Boehmke, Gunter & Schmitz, Gerd, "Polysuccinimide, polyaspartic acid and their salts are prepared by reaction of maleic anhydride and ammonia, polycondensation of the resulting product in the presence of a solubilizing agent and, if appropriate, hydrolysis.", published 1995-11-21, assigned to Bayer AG
- .
- S2CID 84203387.
- PMID 12625712.
- PMID 12161646.
- .
- S2CID 94818855.
- PMID 12161646.
- ^ "Presidential Green Chemistry Challenge Awards: 1996 Small Business Award: Donlar Corporation (now NanoChem Solutions, Inc.): Production and Use of Thermal Polyaspartic Acid". US Environmental Protection Agency.
- PMID 31799235.
- doi:10.1002/pat.1395.
