Condensation polymer
| Polymer science |
|---|
 |

In
Polycondensation A polymerization in which the growth of polymer chains proceeds by condensation reactions between molecules of all degrees of polymerization. Notes: 1. The growth steps are expressed by: Px+Py→Px+y+L {x}∈{1,2,…∞};{y}∈{1,2,…∞}
where Px and Py denote chains of degrees of polymerization x and y, respectively, and L a low-molar-mass by-product. 2. The earlier term 'polycondensation' was synonymous with 'condensation polymerization'. The current definitions of polycondensation and condensative chain polymerization were both embraced by the earlier term 'polycondensation'.[1]
Condensation polymerization is a form of
Polyamides
One important class of condensation polymers are polyamides.[4] They arise from the reaction of carboxylic acid and an amine. Examples include nylons and proteins. When prepared from amino-carboxylic acids, e.g. amino acids, the stoichiometry of the polymerization includes co-formation of water:
- n H2N-X-CO2H → [HN-X-C(O)]n + (n-1) H2O
When prepared from diamines and dicarboxylic acids, e.g. the production of nylon 66, the polymerization produces two molecules of water per repeat unit:
- n H2N-X-NH2 + n HO2C-Y-CO2H → [HN-X-NHC(O)-Y-C(O)]n + (2n-1) H2O

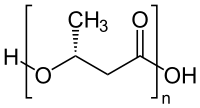
General chemical structure of one type of condensation polymer
Polyesters
Another important class of condensation polymers are
- n HO-X-OH + n HO2C-Y-CO2H → [O-X-O2C-Y-C(O)]n + (2n-1) H2O
Safety and environmental considerations
Condensation polymers tend to be more biodegradable than addition polymers. The peptide or ester bonds between monomers can be hydrolysed, especially in the presence of catalysts or bacterial enzymes.[6]
See also
- Biopolymer
- Epoxy resins
- Polyamide
- Polyester
References
- S2CID 98774337.
- ISBN 0-412-22170-5
- ISBN 978-0-08-011891-8.
- S2CID 241272519.
- ISBN 3-527-30673-0.
- PMID 28371373.

