Propyl gallate
| |
| |
| Names | |
|---|---|
| IUPAC name
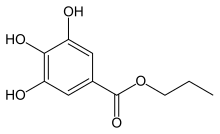
Propyl 3,4,5-trihydroxybenzoate
| |
| Other names
Gallic acid, propyl ester
n-Propyl gallate E310 | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.004.090 |
| EC Number |
|
| E number | E310 (antioxidants, ...) |
| MeSH | Propyl+Gallate |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H12O5 | |
| Molar mass | 212.20 g/mol |
| Appearance | White crystalline powder |
| Melting point | 150 °C (302 °F; 423 K) |
| Boiling point | Decomposes |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Propyl gallate, or propyl 3,4,5-trihydroxybenzoate is an
E310.Description
Propyl gallate is an antioxidant. It protects against oxidation by hydrogen peroxide and oxygen free radicals. It appears as a white to creamy-white crystalline odorless solid.[2][3]
Production
Propyl gallate does not occur naturally, and is prepared either from reactions with
Uses
Propyl gallate is used to protect oils and fats in products from oxidation; it is used in foods, cosmetics, hair products, adhesives, biodiesel, and lubricants.[6] It is often used interchangeably with octyl gallate and dodecyl gallate in these applications.[3]
It is used as a triplet state quencher and an antioxidant in fluorescence microscopy.[7]
Biological effects
A 1993 study in fat rodents found little or no effect on carcinogenesis by propyl gallate.[8]
A 2009 study found that propyl gallate acts as an estrogen antagonist.[9]
References
- S2CID 39562131.
- ISSN 0308-8146.
- ^ .
- ISSN 1381-1177.
- ISSN 1422-8599.
- ISSN 0378-3820.
- PMID 17228891.
- ^ Hirose, Masao, et al.. "Modification of carcinogenesis by α-tocopherol, t-butylhydro-quinone, propyl gallate and butylated hydroxytoluene in a rat multi-organ carcinogenesis model." Carcinogenesis 14.11 (1993): 2359-2364.
- PMID 19063592.
