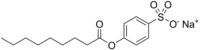
Sodium nonanoyloxybenzenesulfonate
Appearance
| |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium 4-(nonanoyloxy)benzene-1-sulfonate | |
| Other names
4-Sulfophenyl nonanoate sodium salt; Sodium p-nonanoyloxybenzenesulfonate; p-(Nonanoyloxy)benzenesulfonic acid sodium salt; p-Sodiosulfophenyl nonanoate
| |
| Identifiers | |
3D model (
JSmol ) |
|
| Abbreviations | NOBS |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C15H21NaO5S | |
| Molar mass | 336.38 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium nonanoyloxybenzenesulfonate (NOBS) is an important component of
urea peroxide) to effect bleaching at lower temperatures.[1]
Synthesis
NOBS is formed by the reaction of
nonanoic acid (or its esters) with phenol followed by aromatic sulfonation using SO3 to form a sulfonic acid
at the para-position.
Bleach activation
NOBS was developed by Procter & Gamble in 1983[2] and was first used in American laundry detergents in 1988.[3] NOBS is the main bleach activator used in the U.S.A. and Japan.[4] Compared to TAED, which is the predominant bleach activator used in Europe, NOBS is efficient at much lower temperatures. At 20 °C NOBS is 100 times more soluble than TAED in water.[5] When attacked by the perhydroxyl anion (from hydrogen peroxide), NOBS forms the peroxy acid peroxynonanoic acid and releases the leaving group sodium 4-hydroxybenzene sulfonate, which is an inert by-product.
References
- S2CID 235325050.
- ^ Chung, S. Y.; Spadini, G. L. (1983). US4412934.
- ISBN 978-0-935315-52-3.
- ISBN 9780824703516.
- ISBN 9781574447576.
