Solid-state nuclear magnetic resonance

Solid-state NMR (ssNMR) spectroscopy is a technique for characterizing atomic level structure in solid materials e.g. powders, single crystals and amorphous samples and tissues using nuclear magnetic resonance (NMR) spectroscopy. The anisotropic part of many spin interactions are present in solid-state NMR, unlike in solution-state NMR where rapid tumbling motion averages out many of the spin interactions. As a result, solid-state NMR spectra are characterised by larger linewidths than in solution state NMR, which can be utilized to give quantitative information on the molecular structure, conformation and dynamics of the material. Solid-state NMR is often combined with magic angle spinning to remove anisotropic interactions and improve the resolution as well as the sensitivity of the technique.

Nuclear spin interactions
The
Chemical shielding
Chemical shielding is a local property of each nuclear site in a molecule or compound, and is proportional to the applied external magnetic field. The external magnetic field induces currents of the electrons in molecular orbitals. These induced currents create local magnetic fields that lead to characteristic changes in resonance frequency. These changes can be predicted from molecular structure using empirical rules or quantum-chemical calculations.
In general, the chemical shielding is anisotropic because of the anisotropic distribution of molecular orbitals around the nuclear sites. Under sufficiently fast magic angle spinning, or under the effect of molecular tumbling in solution-state NMR, the anisotropic dependence of the chemical shielding is time-averaged to zero, leaving only the isotropic chemical shift.
Dipolar coupling

Nuclear spins exhibit a
- ,
where γ1 and γ2 are the gyromagnetic ratios of the nuclei, is the
- .
D becomes zero for . Consequently, two nuclei with a dipolar coupling vector at an angle of θm = 54.7° to a strong external magnetic field have zero dipolar coupling. θm is called the magic angle. Magic angle spinning is typically used to remove dipolar couplings weaker than the spinning rate.
Quadrupolar interaction
Nuclei with a spin quantum number >1/2 have a non-spherical charge distribution and an associated electric quadrupole moment tensor. The nuclear electric quadrupole moment couples with surrounding electric field gradients. The nuclear quadrupole coupling is one of the largest interactions in NMR spectroscopy, often comparable in size to the Zeeman coupling. When the nuclear quadrupole coupling is not negligible relative to the Zeeman coupling, higher order corrections are needed to describe the NMR spectrum correctly. In such cases, the first-order correction to the NMR transition frequency leads to a strong anisotropic line broadening of the NMR spectrum. However, all symmetric transitions, between and levels are unaffected by the first-order frequency contribution. The second-order frequency contribution depends on the P4 Legendre polynomial, which has zero points at 30.6° and 70.1°. These anisotropic broadenings can be removed using DOR (DOuble angle Rotation) where you spin at two angles at the same time, or DAS (Double Angle Spinning)[2] where you switch quickly between the two angles. Both techniques were developed in the late 1980s, and require specialized hardware (probe). Multiple quantum magic angle spinning (MQMAS) NMR was developed in 1995 and has become a routine method for obtaining high resolution solid-state NMR spectra of quadrupolar nuclei.[3][4] A similar method to MQMAS is satellite transition magic angle spinning (STMAS) NMR developed in 2000.
J-coupling
The
Other interactions
Paramagnetic substances are subject to the Knight shift.
Solid-state NMR line shapes
Powder pattern

A powder pattern arises in powdered samples where crystallites are randomly oriented relative to the magnetic field so that all molecular orientations are present. In presence of a chemical shift anisotropy interaction, each orientation with respect to the magnetic field gives a different resonance frequency. If enough crystallites are present, all the different contributions overlap continuously and lead to a smooth spectrum.
Fitting of the pattern in a static ssNMR experiment gives information about the shielding tensor, which are often described by the isotropic chemical shift , the chemical shift anisotropy parameter , and the asymmetry parameter .[5]
Dipolar pattern
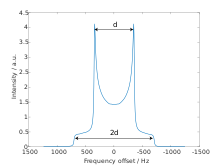
The dipolar powder pattern (also Pake pattern) has a very characteristic shape that arises when two nuclear spins are coupled together within a crystallite. The splitting between the maxima (the "horns") of the pattern is equal to the dipolar coupling constant .:[6]
where γ1 and γ2 are the gyromagnetic ratios of the dipolar-coupled nuclei, is the internuclear distance, is the
Essential solid-state techniques
Magic angle spinning

Magic angle spinning (MAS) is a technique routinely used in solid-state NMR to produce narrower NMR and more intense NMR lines. This is achieved by rotating the sample at the
Spinning a powder sample at a slower rate than the largest component of the chemical shift anisotropy results in an incomplete averaging of the interaction, and produces a set of spinning sidebands in addition to the isotropic line, centred at the isotropic chemical shift. Spinning sidebands are sharp lines separated from the isotropic frequency by a multiple of the spinning rate. Although spinning sidebands can be used to measure anisotropic interactions, they are often undesirable and removed by spinning the sample faster or by recording the data points synchronously with the rotor period.
Cross-polarisation

Cross-polarization (CP) if a fundamental RF pulse sequence and a building-block in many solid-state NMR. It is typically used to enhance the signal of a dilute nuclei with a low gyromagnetic ratio (e.g. 13
C, 15
N) by magnetization transfer from an abundant nuclei with a high gyromagnetic ratio (e.g. 1
H), or as a spectral editing method to get through space information (e.g. directed 15
N→13
C CP in protein spectroscopy).
To establish magnetization transfer, RF pulses ("contact pulses") are simultaneously applied on both frequency channels to produce fields whose strength fulfil the Hartmann–Hahn condition:[7][8]
where are the gyromagnetic ratios, is the spinning rate, and is an integer. In practice, the pulse power, as well as the length of the contact pulse are experimentally optimised. The power of one contact pulse is typically ramped to achieve a more broadband and efficient magnetisation transfer.
Decoupling
Spin interactions can be removed (decoupled) to increase the resolution of NMR spectra during the detection, or to extend the lifetime of the nuclear magnetization.
Heteronuclear decoupling is achieved by radio-frequency irradiation on at the frequency of the nucleus to be decoupled, which is often 1H. The irradiation can be continuous (continuous wave decoupling[9]), or a series of pulses that extend the performance and the bandwidth of the decoupling (TPPM,[10] SPINAL-64,[11] SWf-TPPM[12])
Homonuclear decoupling is achieved with multiple-pulse sequences (WAHUHA,[13] MREV-8,[14] BR-24,[15] BLEW-12,[16] FSLG[17]), or continuous wave modulation (DUMBO,[18] eDUMBO[19]). Dipolar interactions can also be removed with magic angle spinning. Ultra fast MAS (from 60 kHz up to above 111 kHz) is an efficient way to average all dipolar interactions, including 1H–1H homonuclear dipolar interactions, which extends the resolution of 1H spectra and enables the usage of pulse sequences used in solution state NMR.[20][21]
Advanced solid-state NMR spectroscopy
Rotational Echo DOuble Resonance (REDOR)

Rotational Echo DOuble Resonance (REDOR) experiment,
Ultra Fast MAS for 1H NMR
The strong 1H-1H homonuclear dipolar interactions associated with broad NMR lines and short T2 relaxation time effectively relegate proton for bimolecular NMR. Recent developments of faster MAS, and reduction of dipolar interactions by deuteration have made proton ssNMR as versatile as in solution. This includes spectral dispersion in multi-dimensional experiments[24] as well as structurally valuable restraints and parameters important for studying material dynamics.[25]
Ultra-fast NMR and the associated sharpening of the NMR lines enables NMR pulse sequences to capitalize on proton-detection to improve the sensitivity of the experiments compared to the direct detection of a spin-1/2 system (X). Such enhancement factor is given by:
where are the gyromagnetic ratios, represent the NMR line widths, and represent the quality factor of the probe resonances.[26]
MAS-Dynamic Nuclear Polarisation (MAS-DNP)
Magic angle spinning dynamic nuclear polarization (MAS-DNP) is a technique that increases the sensitivity of NMR experiments by several orders of magnitude.[27][28] It involves the transfer of the very high electron polarisation from unpaired electrons to nearby nuclei. This is achieved at cryogenic temperatures by the means of a continuous microwave irradiation coming from a klystron or a gyrotron, with a frequency close to the corresponding electron paramagnetic resonance (EPR) frequency.
The development in the MAS-DNP instrumentation, as well as the improvement of polarising agents (TOTAPOL, AMUPOL, TEKPOL, etc.[29]) to achieve a more efficient transfer of polarisation has dramatically reduced experiments times which enabled the observation of surfaces,[30] insensitive isotopes,[31] and multidimensional experiments on low natural abundance nuclei,[32] and diluted species.[33]
Applications
Solid-state NMR spectroscopy serves as an effective analytical tool in biological, organic, and inorganic chemistry due to its close resemblance to liquid-state spectra while providing additional insights into anisotropic interactions.[34]
It is used to characterize chemical composition, supramolecular structure, local motions, kinetics, and thermodynamics, with the special ability to assign the observed behavior to specific sites in a molecule. It is also crucial in the area of surface and interfacial chemistry.[35]
Biology and Medicine
Proteins and bioaggregates
Solid-state NMR is used to study insoluble proteins and proteins very sensitive to their environment such as
Biomaterials
Solid-state NMR has also been successfully used to study biomaterials such as
Drugs and drug delivery systems
Other ssNMR applications are in the field of pharmaceutical research.[55]
Firstly, it plays a pivotal role in the characterizatio of drug polymorphs and solid dispersions. This is useful to minimize risks linked with solid-state form changes: the determination of the solid form of an
Furthermore, ssNMR aids in characterizing porous materials tailored for drug delivery systems, thus enhabling scientists to design carriers and formulations that heighten drug efficacy. [56]
Materials science
Solid-state NMR has been successfully used to study metal organic frameworks (MOFS),[57] batteries,[58][59][60] surfaces of nanoporous materials,[61] polymers.[62]
Art conservation
NMR can also be applied to art conservation. Different salts and moisture levels can be detected through the use of solid state NMR. However, sampling sizes retrieved from works of art in order to run through these large conducting magnets typically exceed levels deemed acceptable. Unilateral NMR techniques use portable magnets that are applied to the object of interest, bypassing the need for sampling.[63]
References
- ^ "National Ultrahigh-Field NMR Facility for Solids". Retrieved 2014-09-22.
- PMID 17638585.
- .
- PMID 8925268.
- OCLC 53178681.
- OCLC 53178681.
- .
- .
- ISSN 0031-899X.
- ISSN 0021-9606.
- PMID 10617439.
- .
- ISSN 0031-9007.
- ISSN 0021-9606.
- .
- .
- .
- .
- S2CID 203139911.
- PMID 28353338.
- PMID 31891271.
- ^
Gullion T.; Schaefer J. (1989). "Rotational-echo double-resonance NMR". J. Magn. Reson. 81 (2): 196–200. PMID 22152360.
- ISBN 978-0-470-03459-0, retrieved 2021-09-13
- ^
Linser R.; Fink U.; Reif B. (2008). "Proton-Detected Scalar Coupling Based Assignment Strategies in MAS Solid-State NMR Spectroscopy Applied to Perdeuterated Proteins". J. Magn. Reson. 193 (1): 89–93. PMID 18462963.
- PMID 20977205.
- PMID 29223566.
- PMID 29157490.
- S2CID 189814925.
- S2CID 189814925.
- PMID 23517009.
- PMID 26098846.
- S2CID 199000068.
- PMID 24410528.
- .
- . Retrieved 20 December 2023.
- PMID 19245337.
- PMID 21219138.
- ^ Demers, Jean-Philippe; Fricke, Pascal; Shi, Chaowei; Chevelkov, Veniamin; Lange, Adam (December 2018). "Structure determination of supra-molecular assemblies by solid-state NMR: Practical considerations". Progress in Nuclear Magnetic Resonance Spectroscopy. 109: 51–78. Retrieved 20 December 2023.
- S2CID 26146114.
- )
- PMID 25797009.
- S2CID 6854002.
- ISSN 2673-4915.
- S2CID 95774717.
- ISSN 0079-6565.
- ISBN 978-0-12-505337-2, retrieved 2021-09-13
- PMID 25919197.
- PMID 33177698.
- PMID 26355148.
- S2CID 215409770.
- PMID 22331896.
- PMID 22298470.
- S2CID 19939314.
- PMID 25892587.
- .
- .
- PMC 5449066.
- PMID 24041242.
- S2CID 47012638.
- PMID 26974032.
- ISSN 0897-4756.
- OCLC 31785818.
- S2CID 206902463.
Suggested readings for beginners
General NMR
- Keeler, James (2002). "Understanding NMR Spectroscopy". Apollo-University Of Cambridge Repository, Apollo-University Of Cambridge Repository. doi:10.17863/CAM.968.
- Hore, P. J. (2015). NMR : the toolkit : how pulse sequences work. J. A. Jones, Stephen Wimperis (2nd ed.). Oxford. )
- Hore, P. J. (2015). Nuclear magnetic resonance (2nd ed.). Oxford. )
- Encyclopedia of NMR. Robin K. Harris, Roderick E. Wasylishen. Chichester, West Sussex: John Wiley & Sons. 2012. )
Solid-state NMR
- Laws David D., Hans- , Bitter Marcus L., Jerschow Alexej (2002). "Solid-State NMR Spectroscopic Methods in Chemistry". Angewandte Chemie International Edition. 41 (17): 3096–3129. PMID 12207374.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - Reif, Bernd; Ashbrook, Sharon E.; Emsley, Lyndon; Hong, Mei (2021). "Solid-state NMR spectroscopy". Nature Reviews Methods Primers. 1. PMID 34368784.
- Levitt, Malcolm H., Spin Dynamics: Basics of Nuclear Magnetic Resonance, Wiley, Chichester, United Kingdom, 2001. ISBN 978-0470511176(NMR basics, including solids)
- Duer, Melinda J., Introduction to Solid-State NMR Spectroscopy, Blackwell, Oxford, 2004. (Some detailed examples of ssNMR spectroscopy)
- Schmidt-Rohr, K. and Spiess, H.-W., Multidimensional Solid-State NMR and Polymers, Academic Press, San Diego, 1994.
External links
- mrsimulator Python package for simulating solid-state NMR spectra.
- SSNMRBLOG Solid-State NMR Literature Blog by Prof. Rob Schurko's Solid-State NMR group at the University of Windsor
- "Solid-State MAS NMR | Protein NMR". Retrieved 2021-09-13.
- "University of Ottawa NMR Facility Blog". u-of-o-nmr-facility.blogspot.com. Retrieved 2021-09-13.





















