Infrared spectroscopy

Infrared spectroscopy (IR spectroscopy or vibrational spectroscopy) is the measurement of the interaction of
The infrared portion of the electromagnetic spectrum is usually divided into three regions; the near-, mid- and far- infrared, named for their relation to the visible spectrum. The higher-energy near-IR, approximately 14,000–4,000 cm−1 (0.7–2.5 μm wavelength) can excite overtone or combination modes of molecular vibrations. The mid-infrared, approximately 4,000–400 cm−1 (2.5–25 μm) is generally used to study the fundamental vibrations and associated rotational–vibrational structure. The far-infrared, approximately 400–10 cm−1 (25–1,000 μm) has low energy and may be used for rotational spectroscopy and low frequency vibrations. The region from 2–130 cm−1, bordering the microwave region, is considered the terahertz region and may probe intermolecular vibrations.[1] The names and classifications of these subregions are conventions, and are only loosely based on the relative molecular or electromagnetic properties.
Uses and applications


Infrared spectroscopy is a simple and reliable technique widely used in both organic and inorganic chemistry, in research and industry. In catalysis research it is a very useful tool to characterize the catalyst,[2][3][4] as well as to detect intermediates[5] and products during the catalytic reaction. It is used in quality control, dynamic measurement, and monitoring applications such as the long-term unattended measurement of CO2 concentrations in greenhouses and growth chambers by infrared gas analyzers.[citation needed]
It is also used in
IR spectroscopy has been successfully used in analysis and identification of
Infrared spectroscopy is also useful in measuring the degree of polymerization in polymer manufacture. Changes in the character or quantity of a particular bond are assessed by measuring at a specific frequency over time. Modern research instruments can take infrared measurements across the range of interest as frequently as 32 times a second. This can be done whilst simultaneous measurements are made using other techniques. This makes the observations of chemical reactions and processes quicker and more accurate.[citation needed]
Infrared spectroscopy has also been successfully utilized in the field of semiconductor microelectronics:[9] for example, infrared spectroscopy can be applied to semiconductors like silicon, gallium arsenide, gallium nitride, zinc selenide, amorphous silicon, silicon nitride, etc.
Another important application of infrared spectroscopy is in the food industry to measure the concentration of various compounds in different food products.[10][11]
The instruments are now small, and can be transported, even for use in field trials.[citation needed]
Infrared spectroscopy is also used in gas leak detection devices such as the DP-IR and EyeCGAs.[12] These devices detect hydrocarbon gas leaks in the transportation of natural gas and crude oil.
In February 2014,
Infrared spectroscopy is an important analysis method in the recycling process of household
Other developments include a miniature IR-spectrometer that's linked to a cloud based database and suitable for personal everyday use,[16] and NIR-spectroscopic chips[17] that can be embedded in smartphones and various gadgets.
Infrared spectroscopy coupled with machine learning and artificial intelligence also has potential for rapid, accurate and non-invasive sensing of bacteria.[18] The complex chemical composition of bacteria, including nucleic acids, proteins, carbohydrates and fatty acids, results in high-dimensional datasets where the essential features are effectively hidden under the total spectrum. Extraction of the essential features therefore requires advanced statistical methods such as machine learning and deep-neural networks. The potential of this technique for bacteria classification have been demonstrated for differentiation at the genus,[19] species[20] and serotype[21] taxonomic levels, and it has also been shown promising for antimicrobial susceptibility testing,[22][23][24] which is important for many clinical settings where faster susceptibility testing would decrease unnecessary blind-treatment with broad-spectrum antibiotics. The main limitation of this technique for clinical applications is the high sensitivity to technical equipment and sample preparation techniques, which makes it difficult to construct large-scale databases. Attempts in this direction have however been made by Bruker with the IR Biotyper for food microbiology.[25]
Theory

Infrared spectroscopy exploits the fact that molecules absorb frequencies that are characteristic of their
Number of vibrational modes
In order for a vibrational mode in a sample to be "IR active", it must be associated with changes in the molecular dipole moment. A permanent dipole is not necessary, as the rule requires only a change in dipole moment.[26]
A molecule can vibrate in many ways, and each way is called a vibrational mode. For molecules with N number of atoms, geometrically

Simple
The atoms in a CH2X2 group, commonly found in organic compounds and where X can represent any other atom, can vibrate in nine different ways. Six of these vibrations involve only the CH2 portion: two stretching modes (ν): symmetric (νs) and antisymmetric (νas); and four bending modes: scissoring (δ), rocking (ρ), wagging (ω) and twisting (τ), as shown below. Structures that do not have the two additional X groups attached have fewer modes because some modes are defined by specific relationships to those other attached groups. For example, in water, the rocking, wagging, and twisting modes do not exist because these types of motions of the H atoms represent simple rotation of the whole molecule rather than vibrations within it. In case of more complex molecules, out-of-plane (γ) vibrational modes can be also present.[27]
Symmetry Direction |
Symmetric | Antisymmetric |
|---|---|---|
| Radial | 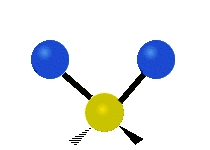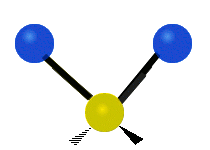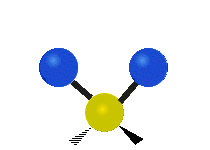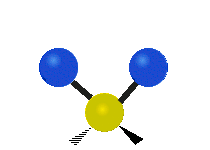 Symmetric stretching (νs) |
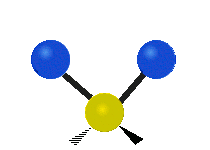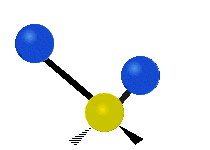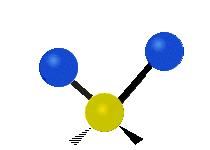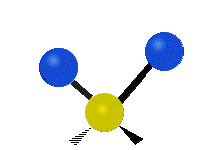 Antisymmetric stretching (νas) |
| Latitudinal |  Scissoring (δ) |
 Rocking (ρ) |
| Longitudinal |  Wagging (ω) |
 Twisting (τ) |
These figures do not represent the "recoil" of the C atoms, which, though necessarily present to balance the overall movements of the molecule, are much smaller than the movements of the lighter H atoms.
The simplest and most important or fundamental IR bands arise from the excitations of normal modes, the simplest distortions of the molecule, from the ground state with vibrational quantum number v = 0 to the first excited state with vibrational quantum number v = 1. In some cases, overtone bands are observed. An overtone band arises from the absorption of a photon leading to a direct transition from the ground state to the second excited vibrational state (v = 2). Such a band appears at approximately twice the energy of the fundamental band for the same normal mode. Some excitations, so-called combination modes, involve simultaneous excitation of more than one normal mode. The phenomenon of Fermi resonance can arise when two modes are similar in energy; Fermi resonance results in an unexpected shift in energy and intensity of the bands etc.[citation needed]
Practical IR spectroscopy
The infrared spectrum of a sample is recorded by passing a beam of infrared light through the sample. When the frequency of the IR is the same as the vibrational frequency of a bond or collection of bonds, absorption occurs. Examination of the transmitted light reveals how much energy was absorbed at each frequency (or wavelength). This measurement can be achieved by scanning the wavelength range using a monochromator. Alternatively, the entire wavelength range is measured using a Fourier transform instrument and then a transmittance or absorbance spectrum is generated using a dedicated procedure.[citation needed]
This technique is commonly used for analyzing samples with covalent bonds. Simple spectra are obtained from samples with few IR active bonds and high levels of purity. More complex molecular structures lead to more absorption bands and more complex spectra.[citation needed]

Sample preparation
Gas samples
Gaseous samples require a sample cell with a long
Liquid samples
Liquid samples can be sandwiched between two plates of a salt (commonly sodium chloride, or common salt, although a number of other salts such as potassium bromide or calcium fluoride are also used).[28] The plates are transparent to the infrared light and do not introduce any lines onto the spectra. With increasing technology in computer filtering and manipulation of the results, samples in solution can now be measured accurately (water produces a broad absorbance across the range of interest, and thus renders the spectra unreadable without this computer treatment).[citation needed]
Solid samples
Solid samples can be prepared in a variety of ways. One common method is to crush the sample with an oily
In photoacoustic spectroscopy the need for sample treatment is minimal. The sample, liquid or solid, is placed into the sample cup which is inserted into the photoacoustic cell which is then sealed for the measurement. The sample may be one solid piece, powder or basically in any form for the measurement. For example, a piece of rock can be inserted into the sample cup and the spectrum measured from it.[citation needed]
A useful way of analyzing solid samples without the need for cutting samples uses ATR or attenuated total reflectance spectroscopy. Using this approach, samples are pressed against the face of a single crystal. The infrared radiation passes through the crystal and only interacts with the sample at the interface between the two materials.[citation needed]
Comparing to a reference
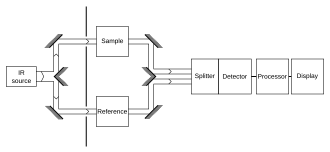
It is typical to record spectrum of both the sample and a "reference". This step controls for a number of variables, e.g. infrared detector, which may affect the spectrum. The reference measurement makes it possible to eliminate the instrument influence.[citation needed]
The appropriate "reference" depends on the measurement and its goal. The simplest reference measurement is to simply remove the sample (replacing it by air). However, sometimes a different reference is more useful. For example, if the sample is a dilute solute dissolved in water in a beaker, then a good reference measurement might be to measure pure water in the same beaker. Then the reference measurement would cancel out not only all the instrumental properties (like what light source is used), but also the light-absorbing and light-reflecting properties of the water and beaker, and the final result would just show the properties of the solute (at least approximately).[citation needed]
A common way to compare to a reference is sequentially: first measure the reference, then replace the reference by the sample and measure the sample. This technique is not perfectly reliable; if the infrared lamp is a bit brighter during the reference measurement, then a bit dimmer during the sample measurement, the measurement will be distorted. More elaborate methods, such as a "two-beam" setup (see figure), can correct for these types of effects to give very accurate results. The Standard addition method can be used to statistically cancel these errors.
Nevertheless, among different absorption-based techniques which are used for gaseous species detection, Cavity ring-down spectroscopy (CRDS) can be used as a calibration-free method. The fact that CRDS is based on the measurements of photon life-times (and not the laser intensity) makes it needless for any calibration and comparison with a reference [29]
Some instruments also automatically identify the substance being measured from a store of thousands of reference spectra held in storage.
FTIR
An alternate method for acquiring spectra is the "dispersive" or "scanning
Infrared microscopy
Various forms of
Other methods in molecular vibrational spectroscopy
Infrared spectroscopy is not the only method of studying molecular vibrational spectra. Raman spectroscopy involves an inelastic scattering process in which only part of the energy of an incident photon is absorbed by the molecule, and the remaining part is scattered and detected. The energy difference corresponds to absorbed vibrational energy.[citation needed]
The selection rules for infrared and for Raman spectroscopy are different at least for some molecular symmetries, so that the two methods are complementary in that they observe vibrations of different symmetries.[citation needed]
Another method is electron energy loss spectroscopy (EELS), in which the energy absorbed is provided by an inelastically scattered electron rather than a photon. This method is useful for studying vibrations of molecules adsorbed on a solid surface.
Recently, high-resolution EELS (HREELS) has emerged as a technique for performing vibrational spectroscopy in a transmission electron microscope (TEM).[33] In combination with the high spatial resolution of the TEM, unprecedented experiments have been performed, such as nano-scale temperature measurements,[34][35] mapping of isotopically labeled molecules,[36] mapping of phonon modes in position- and momentum-space,[37][38] vibrational surface and bulk mode mapping on nanocubes,[39] and investigations of polariton modes in van der Waals crystals.[40] Analysis of vibrational modes that are IR-inactive but appear in
Computational infrared microscopy
By using computer simulations and normal mode analysis it is possible to calculate theoretical frequencies of molecules.[42]
Absorption bands
IR spectroscopy is often used to identify structures because functional groups give rise to characteristic bands both in terms of intensity and position (frequency). The positions of these bands are summarized in correlation tables as shown below.
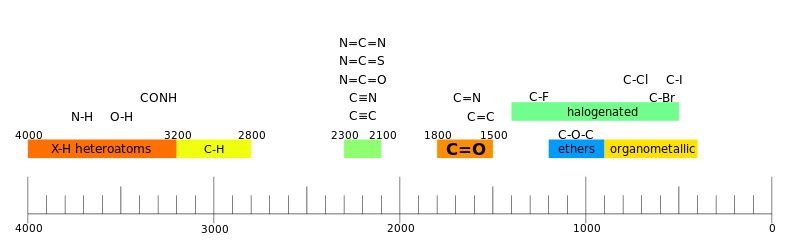
Regions
A spectrograph is often interpreted as having two regions.[43]
- functional group region
In the functional region there are one to a few troughs per functional group.[43]
- fingerprint region
In the fingerprint region there are many troughs which form an intricate pattern which can be used like a fingerprint to determine the compound.[43]
Badger's rule
For many kinds of samples, the assignments are known, i.e. which bond deformation(s) are associated with which frequency. In such cases further information can be gleaned about the strength on a bond, relying on the empirical guideline called Badger's rule. Originally published by Richard McLean Badger in 1934,[44] this rule states that the strength of a bond (in terms of force constant) correlates with the bond length. That is, increase in bond strength leads to corresponding bond shortening and vice versa.
Isotope effects
The different isotopes in a particular species may exhibit different fine details in infrared spectroscopy. For example, the O–O stretching frequency (in reciprocal centimeters) of oxyhemocyanin is experimentally determined to be 832 and 788 cm−1 for ν(16O–16O) and ν(18O–18O), respectively.
By considering the O–O bond as a spring, the frequency of absorbance can be calculated as a wavenumber [= frequency/(speed of light)]
where k is the spring constant for the bond, c is the speed of light, and μ is the reduced mass of the A–B system:
( is the mass of atom ).
The reduced masses for 16O–16O and 18O–18O can be approximated as 8 and 9 respectively. Thus
The effect of isotopes, both on the vibration and the decay dynamics, has been found to be stronger than previously thought. In some systems, such as silicon and germanium, the decay of the anti-symmetric stretch mode of interstitial oxygen involves the symmetric stretch mode with a strong isotope dependence. For example, it was shown that for a natural silicon sample, the lifetime of the anti-symmetric vibration is 11.4 ps. When the isotope of one of the silicon atoms is increased to 29Si, the lifetime increases to 19 ps. In similar manner, when the silicon atom is changed to 30Si, the lifetime becomes 27 ps.[45]
Two-dimensional IR
Two-dimensional infrared correlation spectroscopy analysis combines multiple samples of infrared spectra to reveal more complex properties. By extending the spectral information of a perturbed sample, spectral analysis is simplified and resolution is enhanced. The 2D synchronous and 2D asynchronous spectra represent a graphical overview of the spectral changes due to a perturbation (such as a changing concentration or changing temperature) as well as the relationship between the spectral changes at two different wavenumbers.[citation needed]

Nonlinear two-dimensional infrared spectroscopy
As with two-dimensional nuclear magnetic resonance (
See also
- Applied spectroscopy
- Astrochemistry
- Atomic and molecular astrophysics
- Atomic force microscopy based infrared spectroscopy (AFM-IR)
- Cosmochemistry
- Far-infrared astronomy
- Forensic chemistry
- Forensic engineering
- Forensic polymer engineering
- Infrared astronomy
- Infrared microscopy
- Infrared multiphoton dissociation
- Infrared photodissociation spectroscopy
- Infrared spectroscopy correlation table
- Infrared spectroscopy of metal carbonyls
- Near-infrared spectroscopy
- Nuclear resonance vibrational spectroscopy
- Photothermal microspectroscopy
- Raman spectroscopy
- Rotational-vibrational spectroscopy
- Time-resolved spectroscopy
- Vibrational spectroscopy of linear molecules
References
- S2CID 34705104.
- ISSN 0926-3373.
- S2CID 236149569.
- S2CID 232627637.
- ISSN 0926-3373.
- ^ "Infrared Spectroscopy". ColourLex. 12 March 2021. Retrieved 19 January 2022.
- ISBN 0-89236-469-6.
- ^ Ricciardi P. "Unlocking the secrets of illuminated manuscripts". Retrieved 11 December 2015.
- ISBN 978-981-02-2352-6.
- ISBN 9780470027318.
- PMID 22980869.
- ^ www.TRMThemes.com, TRM Theme by. "Infrared (IR) / Optical Based Archives - Heath Consultants". Heath Consultants. Archived from the original on 2022-01-20. Retrieved 2016-04-12.
- ^ "NASA Ames PAH IR Spectroscopic Database". The Astrophysics & Astrochemistry Laboratory. 12 September 2019. Retrieved 19 January 2022.
- ^ Hoover R (February 21, 2014). "Need to Track Organic Nano-Particles Across the Universe? NASA's Got an App for That". NASA. Archived from the original on September 6, 2015. Retrieved February 22, 2014.
- PMID 30965736.
- ^ Perry, Tekla S. (2017-03-14). "What Happened When We Took the SCiO Food Analyzer Grocery Shopping". IEEE Spectrum: Technology, Engineering, and Science News. Retrieved 2017-03-23.
- ^ Coates, John (18 June 2014). "A Review of New Small-Scale Technologies for Near Infrared Measurements". American Pharmaceutical Review. Coates Consulting LLC. Retrieved 2017-03-23.
- PMID 9551657.
- PMID 1710644.
- PMID 7887620.
- PMID 30015261.
- PMID 30916881.
- PMID 34522918.
- PMID 35248858.
- ^ "IR Biotyper® for Food Microbiology". www.bruker.com. Retrieved 2024-03-20.
- ISBN 0-7167-3539-3.|quote=the electric dipole moment of the molecule must change when the atoms are displaced relative to one another
- ISBN 978-3-527-26446-9.
- ^ ISBN 978-0-632-02017-1.
- S2CID 123834102.
- ^ a b Chromatography/Fourier transform infrared spectroscopy and its applications, by Robert White, p7
- ISBN 9780470027318.
- ^ H M Pollock and D A Smith, The use of near-field probes for vibrational spectroscopy and photothermal imaging, in Handbook of vibrational spectroscopy, J.M. Chalmers and P.R. Griffiths (eds), John Wiley & Sons Ltd, Vol. 2, pp. 1472 - 1492 (2002)
- ^ S2CID 4467249.
- PMID 29547334.
- S2CID 206748146.
- PMID 30705191.
- PMID 29951584.
- S2CID 118999071.
- S2CID 4459728.
- PMID 28733660.
- S2CID 119452520.
- PMID 32271575.
- ^ ISBN 978-0-07-337562-5. Archived from the original(Book) on 2018-06-28. Retrieved 2018-06-30.
- .
- PMID 16803320.
- .
- S2CID 31230696.
- PMID 15212548.









