Syringetin
| |
| Names | |
|---|---|
| IUPAC name
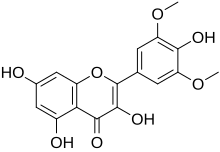
3,4′,5,7-Tetrahydroxy-3′,5′-dimethoxyflavone
| |
| Systematic IUPAC name
3,5,7-Trihydroxy-2-(4-hydroxy-3,5-dimethoxyphenyl)-4H-1-benzopyran-4-one | |
| Other names
3′,5′-O-Dimethylmyricetin
3′,5′-Dimethoxy-3,5,7,4′-tetrahydroxyflavone 3,5,7,4′-Tetrahydroxy-3′,5′-dimethoxyflavone | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C17H14O8 | |
| Molar mass | 346.291 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Syringetin is an
phenolic compounds present in wine.[4]
It induces human osteoblast differentiation through bone morphogenetic protein-2/extracellular signal-regulated kinase 1/2 pathway.[4]
Metabolism
Syringetin is formed from laricitrin by the action of the enzyme laricitrin 5′-O-methyltransferase[1][5] (myricetin O-methyltransferase).[6]
Glycosides
- Syringetin-3-O-galactoside[1][7]
- Syringetin-3-O-glucoside[8][9]
- Syringetin 3-rhamnoside (CAS number 93126-00-2)
- Syringetin-3-O-rutinoside[9] found in Larix sibirica[10]
- Syringetin 3-O-(6′′-acetyl)-β-glucopyranoside found in Picea abies (Norway spruce)[11]
References
- ^ S2CID 21407928.
- S2CID 85252109.
- S2CID 28304488.
- ^ S2CID 42240173.
- ^ "Laricitrin 5′-O-methyltransferase activity". AmiGO 2. Gene Ontology Consortium. 2009-02-28. Retrieved 2021-04-04.
- ^ Foerster, Hartmut (2006-11-03). "MetaCyc pathway: Syringetin biosynthesis". MetaCyc. SRI International. Retrieved 2021-04-04.
- ^ Matsuda, F.; Suzuki, M.; Sawada, Y. (2016-01-19). "Syringetin-3-O-galactoside; LC-ESI-QTOF; MS2; CE:Ramp 5-60 V; [M+H]+". MassBank. Retrieved 2021-04-04.
- ^ Tohge, T. (2016-01-19). "Syringetin-3-O-glucoside; LC-ESI-QTOF; MS". MassBank. Retrieved 2021-04-04.
- ^ S2CID 95953333.
- S2CID 4819832.
- S2CID 84506810.
