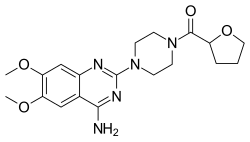
Terazosin
 | |
| Clinical data | |
|---|---|
| Trade names | Hytrin, Zayasel, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a693046 |
| License data | |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 90–94% |
| Elimination half-life | 12 hours |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Terazosin, sold under the brand name Hytrin among others, is a medication used to treat symptoms of an
high blood pressure.[1] For high blood pressure, it is a less preferred option.[1] It is taken by mouth.[1]
Common side effects include dizziness, headache, tiredness, swelling, nausea, and
Terazosin was patented in 1975 and came into medical use in 1985.
Synthesis
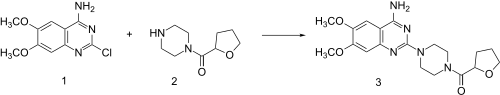
Reaction of
catalytic hydrogenation of the furan
ring leads to 2. This, when heated in the presence of 2-chloro-6,7-dimethoxyquinazolin-4-amine (1) undergoes direct alkylation to terazosin (3).
Research
A 2022 study suggests that terazosin may have the potential to confer neuroprotection upon motor neurons in motor neuron disease, as a result of its ability to activate PGK1.[7]
References
- ^ a b c d e f g "Terazosin Hydrochloride Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. Retrieved 17 March 2019.
- ISBN 9783527607495.
- ISBN 9780857113382.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Terazosin - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- ^ US 4026894, Winn M, Kyncl J, Dunnigan DA, Jones PH, issued 31 May 1977, assigned to Abbott
- PMID 35963713.
