Allylestrenol
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Gestanin, Gestanon, Perselin, Turinal, others |
| Other names | Allyloestrenol; SC-6393; Org AL-25; 3-Deketo-17α-allyl-19-nortestosterone; 17α-Allylestr-4-en-17β-ol; 17α-(Prop-2-en-1-yl)estr-4-en-17β-ol |
| Pregnancy category |
|
Progestin | |
| ATC code | |
| Legal status | |
| Legal status |
|
| Metabolites | • 17α-Allyl-19-NT[3][1][2] |
| Elimination half-life | "Several hours" or 10 hours[4][1][2] |
| Excretion | Urine (as conjugates)[1][2] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Allylestrenol, sold under the brand names Gestanin and Turinal among others, is a
Allylestrenol was first described in 1958 and was introduced for medical use by 1961.[19][20][21][22] It has been marketed widely throughout the world in the past, but today its availability and usage are relatively limited.[23][6][24][25] It remains available in a few European countries and in a number of Asian countries.[23][6][24][25]
Medical uses
Allylestrenol is used in the treatment of
Allylestrenol has been commonly used in Japan at high dosages, typically 50 mg/day but as much as 100 mg/day, to treat BPH in men.[11][28][29][30][31][32][33][34][35][9][10][36][37][38][39][40][41][42][43] Related medications that have similarly been used to treat BPH, particularly in Japan, include chlormadinone acetate, gestonorone caproate, and oxendolone.[33][38] Allylestrenol has also been studied in the treatment of prostate cancer in Japan.[44][28] The medication has been studied as a puberty blocker in the treatment of precocious puberty as well.[45]
Available forms
Allylestrenol is available in the form of 5 mg oral tablets.[12][46][47] It is typically used at a dosage of 5 to 40 mg/day.[46][47] In Japan, a 25 mg allylestrenol oral tablet, under the brand name Perselin, is marketed for the treatment of BPH.[37]
Side effects
Allylestrenol should not be taken by people who are allergic to
Pharmacology
Pharmacodynamics

Progestogenic and off-target activities
Allylestrenol is a
The binding and activity profiles of allylestrenol and its major
| Compound | PR | AR | ER | GR | MR | SHBG | CBG
|
|---|---|---|---|---|---|---|---|
| Allylestrenol | 0 | 0 | 0 | 0 | ? | 1 | ? |
| 17α-Allyl-19-NT | 186 | 5 | 0 | 10 | ? | 3 | ? |
| Notes: Values are percentages (%). Reference CBG. Sources: [3]
| |||||||
Antigonadotropic effects

Similarly to other progestogens, allylestrenol has potent
Other activities
Allylestrenol is not a significant
Pharmacokinetics
Following
Chemistry
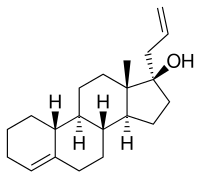
Allylestrenol, also known as 3-deketo-17α-allyl-19-nortestosterone or as 17α-allylestr-4-en-17β-ol, is a
Allylestrenol is also unique among most 19-nortestosterone progestins in that it lacks the
Synthesis
Chemical syntheses of allylestrenol have been published.[58][19][61][62][63]
History
Allylestrenol was patented in 1958
Society and culture
Generic names
Allylestrenol is the
Brand names
The major brand names of allylestrenol include Gestanin, Gestanon, Perselin, and Turinal.[23][6][24][25][19] It has also been marketed under a variety of other brand names, including Alese, Alilestrenol, Allynol, Allytry, Alynol, Anin, Arandal, Astanol, Cobarenol, Crestanon, Elmolan, Fetugard, Foegard, Fulterm, Gestanin, Gestanin, Gestanol, Gestanyn, Gestin, Geston, Gestormone, Gestrenol, Gravida, Gravidin, Gravinol, Gravion, Gravynon, Gynerol, Gynonys, Iugr, Lestron, Loestrol, Maintane, Meieston, Moresafe, Nidagest, Orageston, Pelias, Preabor, Pregnolin, Pregtenol, Pregular, Prelab, Premaston, Prenolin, Prestrenol, Profar, Progeston, Protanon, and Shegest.[23][6][24][25][19]
Availability
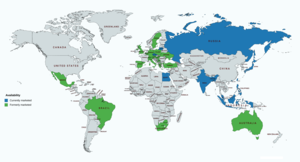
Allylestrenol has been marketed widely throughout the world, including in
References
- ^ ISBN 9780080157450.
- ^ a b c d e f g h i j k l Thijssen JH (1967). Het metabolisme van progestatieve stoffen (Thesis). Rijksuniversiteit te Utrecht.
- ^ PMID 3928974.
- ^ a b Saha A, Roy K, Kakali DE (2000). "Effects of Allylestrenol on Blood Lipids in Relation to its Biological Activity". Indian Journal of Pharmaceutical Sciences. 62 (2): 115.
- ^ a b "SuperCYP".
- ^ ISBN 978-0-85369-840-1.
- ^ PMID 7459930.
- ^ PMID 31745982.
- ^ PMID 1722627.
- ^ PMID 1722628.
- ^ PMID 12534901.
- ^ ISBN 978-3-7692-2114-5.
- ISBN 978-81-312-1158-8.
- ^ ISSN 0804-4643.
- ^ PMID 13699366.
- ^ PMID 13765069.
- ^ S2CID 5612000.
- ^ ISBN 978-0-444-88727-6.
Other examples are allylestrenol (42), a pro-drug converted to the 3-keto analogue (43), which is used in the treatment of threatened abortion [78,79] and altrenogest (44), used in sows and mares to suppress ovulation and estrus behaviour [80]. [...] Progestins with a 17a-allyl side chain: (42) allylestrenol, (43), (44) altrenogest.
- ^ ISBN 978-0-8155-1856-3.
- ^ PMID 13699366.
- ^ ISBN 978-0-19-860027-5.
1961 Lancet 21 Jan. 135/1 Allylestrenol ('Gestanin', Organon)..seems to be completely free from androgenic activity. 1962 Med. Jrnl. Austral. 8 Sept. 375/2 Each tablet of the combined hormone preparation, 'Premenquil', contains 5 mg. of allyloestrenol. [...]
- ^ a b c Medical Proceedings: A South African Journal for the Advancement of Medical Science. Juta and Company. 1962.
Just released in South Africa is Gestanin, Organon Laboratories' new safe oral progestogen. Gestanin is allylestrenol, one of a new group of steroids synthesized by Organon.
- ^ a b c d e f g h i j "Micromedex Products: Please Login".
- ^ a b c d e f g h i j k l m "Allylestrenol".
- ^ ISBN 978-3-88763-075-1.
- PMID 3609436.
- PMID 1473164.
- ^ PMID 6203385.
- PMID 2425610.
- PMID 2425611.
- ^ PMID 2426932.
- ^ PMID 2426933.
- ^ PMID 2435122.
- ^ PMID 1693037.
- ^ PMID 1693038.
- PMID 1384295.
- ^ PMID 7689782.
- ^ PMID 7513937.
- ^ PMID 7541089.
- ^ PMID 9781436.
- PMID 11107528.
- PMID 12094708.
- ^ PMID 16910584.
- S2CID 33940589.
- PMID 5057420.
- ^ ISBN 978-93-5025-937-5.
- ^ ISBN 978-81-7991-527-1.
- ^ a b "Aspirin information from Drugs.com". Archived from the original on 9 May 2008. Retrieved 8 May 2008.
- PMID 16247191.
- PMID 7065053.
- ^ PMID 22078182.
- ISBN 978-1-4832-7088-3.
- ISBN 978-3-540-89760-6.
- ^ PMID 9268084.
- PMID 7000222.
- PMID 6237116.
- PMID 694436.
- ^ ISBN 978-1-4757-2085-3.
- ISBN 978-93-5270-218-3.
- ^ ISBN 978-0-08-093292-7.
- ISBN 978-0471238966.
- ISBN 978-3-642-99941-3.
- ^ De Winter MS, Siegman CM, Szpilfogel SA (1959). "17-alkylated-3-deoxo-19-nor-testosterone". Chem. Ind.: 905.
- ISBN 978-94-011-4439-1.
